NCERT Revision Notes for The p-Block Elements Class 12 Chemistry
CBSE NCERT Revision Notes1
Answer
P – Block of the periodic table comprises six groups (group 13 to 18). The general electronic configuration of the elements present in this block is ns2np1–6 .
All these groups have specific names mostly based on the first member of each group.
- Group 13 : Boron Family
- Group 14 : Carbon Family
- Group 15 : Nitrogen Family
- Group 16 : Oxygen Family
- Group 17 : Halogens
- Group 18 : Noble Gases
With the exception of the noble gases which are very little reactive chemically, the elements belonging to all these groups are reactive. This is the only block in the periodic table which includes all the three types of elements i.e., metals, non – metals and semi – metals. The p – block elements are also known as the Representative Elements.
2
Answer
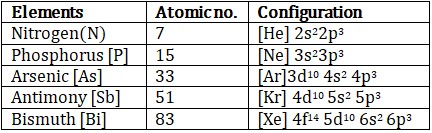
In group 15 of the periodic table, the elements, nitrogen (7N), phosphorus (15P), arsenic (33As), antimony (51Sb) and bismuth (83Bi) are present .
Atomic and Physical Properties
(a) Electronic configuration
The general outer electronic configuration of elements of group 15 is ns2np3. Hence these elements belong to p – block.
(b) Atomic and ionic radii: The atomic (covalent) and ionic radii (in a particular oxidation state) of the elements of nitrogen family (group 15) are smaller than the corresponding elements of carbon family (group 14). On moving down the group, the covalent and ionic radii (in a particular oxidation state) increase with increase in atomic number. There is a considerable increase in covalent radius from N to P. However, from As to Bi, only a small increase is observed. This is due to presence of completely filled d – and/or f – orbitals in heavier members.
(c) Ionisation enthalpy : As the atomic size increase down the group, the ionization enthalpy increases. The ionization enthalpy of nitrogen group element is more than the corresponding elements of oxygen group. This is because more stable half filled outermost p – subshell of nitrogen group elements. Ionisation enthalpy decreases from N to Bi down the group due to gradual increase in atomic size.
(d) Electronegativity : Group 15 elements are more electronegative than group 14 elements. It decreases on moving down the group from N to Bi due to gradual increases in atomic size. N is the 3rd highest in periodic table.
(e) Physical state : Nitrogen is a diatomic gas while other members are solids.
(f) Metallic character : These is increase in metallic character down the group due to increase in atomic size and decrease in ionization enthalpy.
(g) Melting and boiling points : Melting point increases from N to As and then decreases to sb and Bi. Boiling point increase down the group.
(h) Allotropy : All elements except nitrogen show allotropy :
(i) Density : Density increases down the group.
(j) Catenation
They exhibit the property of catenation but due to weak M – M bond to less extent than 14 group elements. ![]()
3
Answer
(a) Oxidation state : The elements of this group can exhibit various oxidation states ranging between −3 to 5. Negative oxidation state will be exhibited when they combine with less electronegative element & positive oxidation state will be exhibited with more electronegative element. Positive oxidation state becomes more favourable as we move down the group due to increasing metallic character & electropositivity. Although due to inert pair effect the stability of +5 state will also decreases. The only stable compound of Bi (v) is BiF5.
(b) Covalency : This is the number of covalent bonds formed by an atom. Maximum covalency of nitrogen is four because it does not have d– orbitals to expand its covalency. Whereas other elements can exhibit covalency of 5 and 6 as well by the use of their d– orbitals e.g. PCl5, [PCl6]−
(c) Reaction with metals : Elements of group 15 react with metals and form binary compounds in –3 oxidation states e.g. Ca3N2
6Li + N2 → 2Li3N ; 6Mg + P4 → 2Mg3P2
(d) Reaction with hydrogen : All elements of group 15 form gaseous hydrides of the type MH3 . In all the hydrides the central atom is sp3 hybridized & their shape is pyramidal duo to presence of lone pair of electrons.
- The basic strength of the hydrides decreases as we move down the group. Thus, NH3 is the strongest base.
NH3 > PH3 > AsH3 > SbH3 - The thermal stability of the hydrides decreases as the atomic size increases, i.e., the M – H bond strength decreases which means reducing character increases.
- In the liquid state, the molecules of NH3 are associated due to hydrogen bonding. The molecules of other hydrides are not associated.
- NH3 is soluble in water whereas other hydrides are insoluble.
- All the hydrides, except NH3, are strong reducing agents and react with metal ions (Ag+ , Cu2+ , etc.) to form phosphides, arsenides or antimonides.
Note : 
(e) Halides : The elements of group 15 form two series of halides MX3 and MX5 .
- All the elements of the group form trihalides. The ionic character of trihalides increases as we move down the group. Except NCl3 , all the trihalides are hydrolysed by water. This is due to the absence of d – orbitals in nitrogen.
- PF3 is not hydrolysed because fluorine being more electronegative than oxygen forms more stable bonds with phosphorus than P – O bonds.
- N cannot form NX5 because of non – availability of d – orbitals. Bi cannot form a BiX5 because of reluctance of 6s electrons of Bi to participate in bond formation.
- The Hybridisation of M in MX3 is sp3 and shape is pyramidal. In MX5 M is sp3d hybridized and shape is trigonal bipyramid. The axial bonds in MX5 are weaker and longer, So MX5 are less stable and decompose on heating
eg: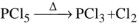
(f) Oxides
- Nitrogen forms a number of oxides. The rest of the members (P, As, Sb and Bi) of the group form two types of oxides; E2O3 and E2O5.
- The reluctance of P, As, Sb and Bi to enter into pπ – pπ multiple bonding leads to cage structures of their oxides and they exist and dimers, E4O6 and E5O10 .
- The basic nature of the oxides increases with increase in atomic number of the element. Thus, the oxides of nitrogen (except N2O and NO), P(III) and As (III) are acidic, Sb (III) oxide is amphoteric and Bi (III) oxide is basic.
4
Answer
The anomalous behaviour of nitrogen is due to its:
(a) Small size
(b) High E.N. and high I.E.
(c) Non availability of vacant d – orbital
(d) Tendency to form multiple bonds.
Nitrogen differs from other elements of its own group in following properties
(a) Nitrogen is a gas while other elements are solids
(b) Nitrogen is diatomic, while other elements are tetratomic [P4, As4, Sb4]
(c) Nitrogen can form N3−ion (due to small size and high E.N.)
(d) Nitrogen is chemically inert under ordinary conditions due to high dissociation energy of N = N bond.
(e)Nitrogen shows oxidation state from –3 to +5
(f) Hydride of nitrogen i.e., ammonia is stable and undergoes H – bonding.
5
Answer
Nitrogen constitutes about 78% by volume of the atmosphere. It occurs as chile saltpetre and Indian saltpetre. It is also an essential constituent of fertilizers, explosives and proteins.
Preparation
(a) Commercially by liquefaction and fractional distillation of air.
(b) In laboratory, by treating and aqueous solution of ammonium chloride with sodium nitrite.
NH4Cl(aq) + NaNo2(aq) → N2(g) + 2H2O(l) + NaCl(aq)
(c) By thermal decomposition of ammonium dichromate and of sodium or barium azide. ![]()
Physical properties
(a) It is a colourless, odourless, tasteless and non – toxic gas.
(b) it has two stable isotopes : 14N and 15N.
(c) It is inert at room temperature because of high bond enthalpy of N ≡ N bond but its reactivity increases rapidly with rise in temperature.
Uses
(a) Used in manufacture of NH3 and other industrial chemicals containing nitrogen, e.g., calcium cyanamide.
(b) As inert diluent for reactive chemicals in iron and steel industry.
(c) As a refrigerant to preserve biological materials, food items and in cryosurgery.
6
Answer
Preparation
(a) Decay of nitrogenous organic matter, e.g., urea. 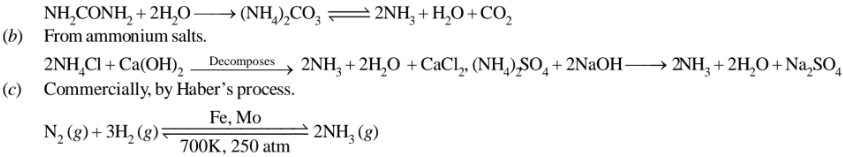
Iron oxide with small amount of K2O and Al2O3 are used as catalysts, at a temperature of 700K and pressure of 200 atm.
Physical Properties
(a) Lighter than air
(b) Easily liquefied by cooling or compression
(c) Highly soluble in water. The solution is alkaline
(d) Forms H – bonding with water.
(e) Colourless gas with pungent odour
Chemical Properties
(a) It is basic and forms ammonium salts with acids.
NH3 + HCl → NH4Cl; 2NH3 + H2SO4 → (NH4)2SO4
(b) Reaction with metal salt ![]()
(c) Reaction with metals
Active metals liberate H2 with NH3 ![]()
(d) Reaction with air
Ammonia burns in presence of catalyst to give NO ![]()
(e) Complex formation ![]()
(f) Oxidation (reducing property)
Uses :
(a) To produce various nitrogenous fertilisers.
(b) Manufacture of some inorganic nitrogen compounds like HNO3 , (NH4)CO3, Na2CO3 etc.
(c) Liquid ammonia is used as a refrigerant.
(d) In making artificial silk and as a laboratory reagent.
Oxides or nitrogen: It forms a number of oxides in different oxidation states – N2O, NO, N2O3, NO2,N2O4, N2O5,
7
Answer
It is colourless neutral gas
Preparation : 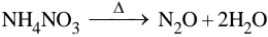
Properties :
(a) Nitrous oxide is a colourless neutral gas with faint pleasant smell. It is heavier than air, fairly soluble in cold water but not in hot water. It is poisonous in nature; when inhaled in moderate quantities, it produces hysterical laughter, hence it is also known as laughing gas.
(b) Action of heat : 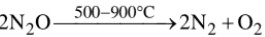
Supporter of Combustion : Although nitrous oxide itself is non – combustible, it supports combustion of glowing splinter, charcoal, burning phosphorus and magnesium ribbon.
Mg + N2O → MgO + N2
Uses :
(a) It is used as the propellant gas in ‘whipped’ cream bombs.
(b) Mixed with oxygen, it is used as an anaesthetic for small scale operations in dentistry and surgery.
Structure: ![]()
Note that N2O is isoelectronic with CO2.
8
Answer
O.S. (+2), colourless gas. This oxide is neutralPreparation
(a) By passing air through an electric arc (Commercial method)
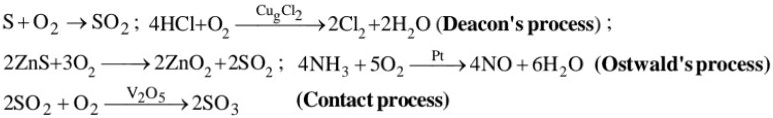
(b) By catalytic oxidation of ammonia (Ostwald method).
(c) 3Cu(s) + 8HNO3(dil) → 3Cu(NO3)2 + 4H2O + 2NO (Lab method)
Properties : It shows oxidizing as well as reducing properties.
Uses
(a) It is used as an intermediate in the manufacture of nitric acid.
(b) It is used as a catalyst in the lead chamber process for the manufacture of H2SO4.
(c) It is used in the detection of oxygen to distinguish it from nitrous oxide.
Structure
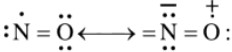
9
Answer
O.S. (+4), brown gas, acidic in nature.
Preparation ![]()
Properties : Highly toxic, paramagnetic, reddish brown gas with choking odour, acidic
Uses :
- For manufacturing of HNO3
- As a catalyst in lead chamber process for sulphuric acid
Structure : 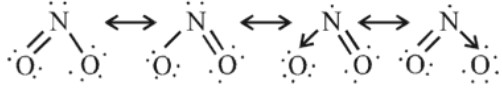
10
Answer
Preparation
(a) 2HNO3 + As2O3 + 2H2O → NO + NO2 + 2H3AsO4
(b) 2Cu + 6HNO3 → 2Cu(NO3)2 + NO + NO2 + 3H2O
Properties :
The brown coloured mixture of NO and NO2 on cooling condenses to a blue liquid which on freezing gives a blue solid of the formula N2O3. 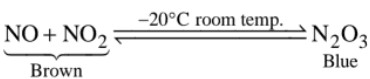
It is an acidic oxide and hence dissolves in alkalies producing nitrites.
N2O3 + 2KOH → 2KNO2 + H2O
Thus dinitrogen trioxide is referred to as the anhydride of nitrous acid.
2HNO2 → N2O3 + H2O
Structure of N2O3 : 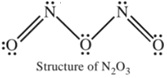
11
Answer
O.S. (+5), colourless gas, acidic in naturePreparation :
4HNO3 + P4O10 → 4H3PO3 + 2N2O5
Properties :
(a) When heated above its m.p. it decomposes with explosion.
(b) N2O5 + H2O → 2HNO3 ,
(c) N2O5 + 2NaOH → 2NaNO3 + H2O
(d) Due to its easy decomposition , it acts as a powerful oxidising agent. It destroys all organic substances like cork, rubber etc.
Structure
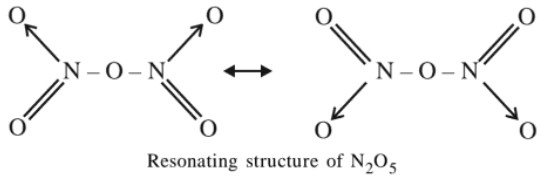
In the vapour state, ionic bond structure changes to covalent structure which is a resonance hybrid of the two resonating structures.
12
Answer
O.S.(+4), colourless solid/liquid, acidic in nature.Preparation :
Structure :
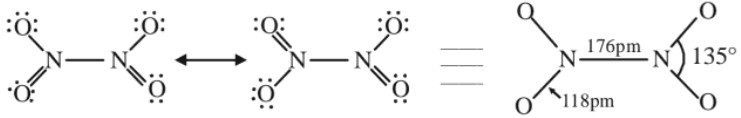
Note : Nitrogen forms oxyacids such as H2N2O2 (hyponitrous acid), HNO2 (nitrous acid) and HNO3 (nitric acid). HNO3 is the most important oxyacid of nitrogen.
13
Answer
Preparation
(a) Lab method: by heating KNO3 or NaNO3 and conc. H2SO4.
NaNO3 + H2SO4 → NaHSO4 +HNO3
(b) Commercially, by Ostwald’s process. ![]()
3NO2(g) + H2O(l) → 2HNO3 (aq) + NO(g)
(c) From air (Birkeland – Eyde electric arc process).
Physical Properties
(a) It is colourless liquid but looks yellow due to dissolved NO2.
(b) It freezes at 231.4 K and boils at 355.6 K.
(c) Fuming HNO3 is pure HNO3 with oxides of nitrogen dissolved in it.
Chemical Properties : ![]()
(b) It is a strong monobasic acid : ![]()
(c) It is a very strong oxidising agent, in concentrated as well as in dilute form :
2HNO3(conc.) → H2O + 2NO2 + [O], 2HNO3(dil.) → H2O + 2NO + 3[O]
(d) Except gold and platinum, HNO3 attacks all metals forming a variety of products.
(e) It oxidises many organic compounds and forms various products on reaction.
(f) Sugar on oxidation with HNO3 gives oxalic acid.
(g) It is strong oxidising agent and attacks most metals except noble metals such as gold and platinum. However, these dissolve in aqua regia (a mixture of 3 parts of conc. HCl and 1 part of conc. HNO3 ).
Uses
(a) Used in manufacture of ammonium nitrate for fertilisers and other nitrates for use in explosives and pyrotechniques.
(b) Preparation of nitroglycerin, trinitrotoluene and other organic nitro compounds.
(c) Pickling of stainless steel, etching of metals and as a oxidizer in rocket fuels.
Structure
Phosphorus
Phosphorus is obtained as phosphates in nature. For e.g. phosphorite (Ca3(PO4)2), chlorapatite hydroxyapatite, Fluoraptite etc.
- Phosphorus occurs in bones, phosphate rocks. It is used in fertilizers.
- As, Sb, Bi, occur mostly as sulphides.
It is isolated by heating Ca3(PO4)2 with coke and silica in an electric furnace. ![]()
Phosphorus exhibits allotropy. The various allotropes of phosphorus are white phosphorus, red phosphorus and black phosphorus. White phosphorus is the most common form whereas black phosphorus is the most stable form of phosphorus.
14
Answer
White phosphorus :(a) It is less stable and more reactive due to angular strain in the P4 molecule where the angles are only 60°.
(b) It readily catches fire in air to give dense fumes of P4O10.
P4 + 5O2 → P4O10
(c) It consists of discrete tetrahedral P4 molecule.
(d) White P burn in oxygen at 30°C forming trioxide and pentoxide.
(e) Reaction with caustic alkalies (NaOH and KOH) gives phosphine and sodium hypophosphite.
(f) White phosphorus react at ordinary temperature and on heating with chlorine it gives PCl3 and PCl5 .
(g) White phosphorus acts as a strong reducing agent. It reduces HNO3 to NO2 , H2SO4 to SO2 and CuSO4 to Cu and itself oxidized to H3PO4.
Red phosphorus:
Red phosphorus is obtained by heating white phosphorus in an inert atmosphere for several days.
(a) It possesses iron grey lustre.
(b) It is odourless, non – poisonous, insoluble in H2O as well as CS2.
(c) Chemically, it is less reactive than white phosphorus.
(d) It does not glow in the dark.
(e) Red phosphorus is belived to exist as chains of P4 tetrahedra linked together.
(f) Red phosphorus does not react with caustic alkalis.
(g) Red phosphorus react at ordinary temp. and on heating respectively with chlorine giving PCl3 and PCl5 .
Black phosphorus
Black phosphorus has two forms : α– and β–
(a) α – black phosphorus is formed by heating red phosphorus. It has opaque monoclinic or rhombohedral crystals. It does not oxidise in air.
(b) β– black phosphorus is prepared by heating white phosphorus under high pressure. It does not burn in air upto 673 K.
15
Answer
Preparation(a) Ca3P2 + 6H2O → 3Ca(OH)2 + 2PH3
(b) Ca3P2 + 6HCl → 3CaCl2 + 2PH3
(c) P4 + 3NaOH + 3H2O → PH3 + 3NaH2PO2
Properties
(a) It is a colourless gas with rotten fish smell and is poisonous.
(b) It is slightly soluble in H2O and is weakly basic, gives phosphonium compounds with acids, e.g. :
PH3 + HBr → PH4Br
(c) The solution of PH3 in water decomposes in presence of light giving red phosphorus and H2.
(d) Phosphine when comes in contact with air, forms vortex rings of P2O5 in the form of white smoke. On the basis of this property.
phosphine is used to prepare smoke screens in warfare. Calcium phosphide reacts with water forming phosphine which burns in air to give clouds or P2O5 which in turn acts as smoke screens.
(e) PH3 is a powerful reducing agent.
Structures :
It has a pyramidal structure with H – P – H angle of 93.6°.
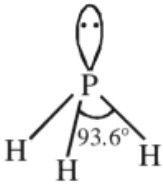
Uses :
(a) It is used in smoke screens.
(b) Containers containing calcium carbide and calcium phosphide are pierced and thrown in the sea when the gases evolved burn and serve as a signal. These are called Home’s signals.
16
Answer
Phosphorus forms two types of halides, PX3 and PX5 (X = F, Cl, Br, l)
PX3 (PCl3)
Preparation
(a) P4 + 6Cl2 → 4PCl3
(b) P4 + 8SOCl2 → 4PCl2 + 4SO2 + 2S2Cl2
Properties
(a) It is colourless oily liquid and undergo hydrolysis in presence of moisture.
PCl3 + 3H2O → H3PO3 + 3HCl
(b) It reacts with organic compounds containing –OH group
3CH3COOH + PCl3 → 3CH3COCl + H3PO3
3C2H5OH + PCl3 → 3C2H5Cl + H3PO3
Structure 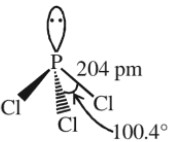
It has a pyramidal shape in which P is sp3 –hybridized.
17
Answer
Preparation(a) P4 + 10Cl2 → 4PCl5
(b) P4 + 10SO2Cl2 → 4PCl5 + 10SO2
Properties
(a) It is a yellowish white powder.
(b) In moist air, it hydrolysis to give POCl3 and finally gets converted to H3PO4.
PCl5 + H2O → POCl3 + 2HCl,
POCl3 + 3H2O → H3PO4 + 3HCl
(c) It sublimes on heating and decomposes on stronger heating
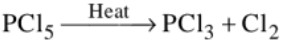
(d) It reacts with organic compounds containing –OH group
C2H5OH + PCl3 → C2H5Cl + POCl3 + HCl, CH3COOH + PCl5 → CH3COCl + POCl3 + HCl
(e) Finely divided metals on heating with PCl5 give corresponding chlorides.
2Ag + PCl5 → 2AgCl + PCl3 , Sn + 2PCl5 → SnCl4 + 2PCl3
Structure :
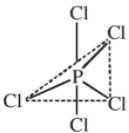
In gaseous and liquid phases, it has a trigonal bipyramidal structure. The three equatorial P –Cl bonds are equivalent, while the two axial bonds are longer than equatorial bonds. This is due to the fact that axial bonds suffer more repulsion than equatorial bonds.
In solid state, it exists as an ionic solid, [PCl4]+ [PCl6]− in which the cation [PCl4]+ is tetrahedral and the anion [PCl4]− is octahedral.
Uses :
It is used in the synthesis of organic compounds like C2H5Cl, CH3COCl etc. as a chlorinating agent.
18
Answer
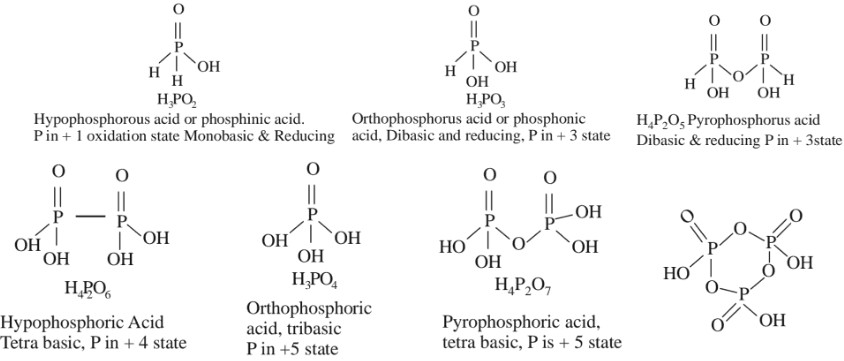
Cyclotrimetaphoshoric acid tribasic, P is +5 state, In all these acids, P – H linkages are reducing & P – OH linkages are acidic, so number of P – OH linkages is the basicity of the acid.
19
Answer
Structures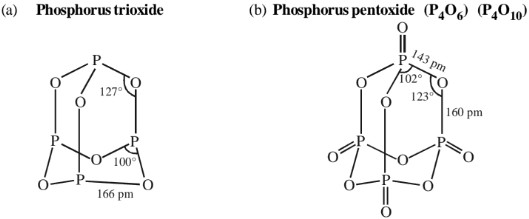
Each phosphorus atom in P4O6 and P4O10 lie at the corners of a tetrahedron. In P4O6 each P is covalently bonded to three O – atoms and each O is bonded to two P – atoms. Thus, the six O – atoms lie along the edges of tetrahedron forming six P – O – P bonds.
In P4 O10 . each P also forms a double bond by sharing its lone pair of electrons with an O – atom.
20
Answer
In group 16 of the periodic table, elements, oxygen (8O), sulphur (16S), selenium (34Se), tellurium (52Te) and polonium (84Po) are present.
Atomic and Physical Properties
(a) Electronic configuration : The elements have the structure ns2np4 for their valence shells. The first element of the group 16 differs in its chemical behaviour from that of other members of the group due to its small size and high electronegativity.
(b) Metallic character : The metallic character increase with increase in atomic number. The first four elements are non – metallic in character. Non – metallic character is strongest in O and S, weaker in Se and Te while Po is metallic.
(c) Atomic and ionic radii : Atomic and ionic radii increases from top to bottom, due to increase in the number of shells.
(d) Ionisation enthalpy : Ionisation enthalpy decreases down the group, due to increase in size. Elements of group 16 have lower ionisation enthalpy values as compared to group 15 in the corresponding periods. This is due to the fact that group 15 elements have extra stable half – filled P – orbital electronic configurations.
(e) Electron gain enthalpy : Oxygen has less negative electron gain enthalpy than sulphur due to compact nature of oxygen atom. From sulphur onwards, electron gain enthalpy becomes less negative upto Po.
(f) Electronegativity : Next to F, O has highest electronegativity value among the elements. Within the group, electronegativity decreases with increase in atomic number.
(g) Catenation : The tendency for catenation decreases markedly as we go down group. Sulphur has a strong tendency for catenation. This is evident from the formation of polysulphided Sn2− , polysulphonic acids, HO3S–Sn–SO3 Hand sulphanes, H–Sn–H .
(h) Density : Increases down the group regularly.
(i) Melting point and boiling point : Both show a regular increase down the group due to increase in molecular weight and Van der Wall’s forces of attraction .
(j) Atomicity : Oxygen is diatomic, sulphur and selenium octa atomic with puckered ring structure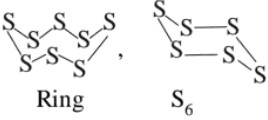
(k) Allotropy : All the elements exhibit allotropy.
Chemical Properties
(a) Oxidation state : Oxygen shows oxidation state of −2 but in case of OF2 and O2F2 oxidation state is +2 and +1 respectively. Sulphur, selenium, tellurium and polonium shows +2, +4 and +6 oxidation state. Stability of +6 oxidation state decreases down the group and +4 oxidation state increases down the group due to inert pair effect. ![]()
In OF2 the oxidation state of oxygen is +2
(b) Reaction with hydrogen : All the elements of the group form volatile hydrides.
- The volatility increases markedly form water to hydrogen sulphide and then declines.
This is evident in their boiling point. Increasing order of boiling points of hydrides is H2S < H2Se < H2Te < H2 Down the group boiling point increases because of increasing molecular weight increases the van der Waal’s interaction. H2O has abnormally high b.p. due to hydrogen bonding. - The thermal stability of the hydrides decreases in the order : H2O > H2S > H2Se > H2Te> H2
- The strength of the hydrides as acids increases in the order: H2O < H2S < H2Se < H2
- The reducing power of the hydrides increases from H2O to H2
(c) Reaction with halogens : Group 16 elements form number of halides of type EX4 and EX6. Where E is an group 16 element and X is a halogen. Stability of halides decrease in the order F > Cl− > Br− > I− .
- Hexahalides – These are formed by fluorine only (not by Cl, Br, I) where elements exhibit maximum valency of +6. SF6, SeF6, TeF6 are colourless gases with sp3d2 hybridisation and octahedral structure. These are covalent in nature. Due to bigger size of Cl, Br and I the coordination number of 6 is not achieved.
- Tetrahalides – With the exception of SBr4, SI4 and SeI4 all tetrahalides are known.
They have trigonal bipyramidal shape with sp3d hybridiation. - Dihalides – The dihalides eg SCl2, OF2, TeBr2 are sp3, hybridised and have distorted bond angles due to electron pair repulsions .
- Dimeric monohalides – The dimeric monohalides are given by sulphur and selenium eg. S2F2, S2Cl2, Se2Cl2, S2Br2, Se2Br2 .
These oxides become less acidic as we go down the group. Reducing property of dioxides decreases from SO2 to TeO2.
S, Se and Te form trioxides of the formula MO3. In the gaseous phase, SO3 exist as a planar triangular molecule.
21
Answer
Preparation
(a) By heating oxygen containing salts, such asa chlorates, nitrates and permanganates. 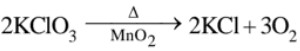
(b) By thermal decomposition of the oxides of metals low in the electrochemical series and higher oxides of some metals.
2Ag2O(s) → 4Ag(s) + O2 (g)
(c) By catalytic decomposition of H2O2.
2H2O2(aq) → 2H2O(l)+ O2(g)
(d) From water by electrolysis. Hydrogen is released at cathode and oxygen at anode.
(e) Industrial preparation : By liquefaction of air followed by its fractional distination. Carbon and water vapour are removed before liquefaction of the air.
Properties :
(a) Colourless and odourless gas soluble in water.
(b) It has three stable isotopes: 16O, 17O and 18O.
(c) It reacts with nearly all metals and non – metals except Au, Pt and some noble gases. Some reactions with metals, non – metals and other compounds are given as:
2Ca + O2 → 2CaO; 2Mg+O2 → 2MgO; 3Fe + 2O2 → Fe3O4 : C + O2 → CO2; P4 + 5O2 → 2P2O5:
22
Answer
Binary compounds of oxygen with another element are called oxides. Oxides are of two types.
(a) Simple oxides – MgO, Al2O3.
(b) Mixed oxides – Fe3O4, Al2O3.
Simple oxides are of four types.
(i) Acidic (Non – metal oxides)
(ii) Basic (Metal oxides)
(iii) Amphoteric
(iv) Neutral
- Oxides which give an acid with water are called acidic oxides. General non – metal are acidic, e.g. SO2, PO5, SO3, I2O5.
Oxides of some metals in high oxidation state are also acidic, e.g., Mn2O7, Cr2O3, V2O5, etc. - Oxides which give a base with water are basic oxides. Generally metal oxides are basic e.g. Na2O, CaO, BaO, etc.
- Oxides which exhibit dual behaviour i.e., show properties of both acidic and basic oxides are called amphoteric oxides, e.g., ZnO, Al2O3 , SnO
- Oxides which are neither acidic nor basic are called neutral oxides, e.g., CO, NO and N2
Uses :
(a) In respiration and combustion processes.
(b) In oxyacetylene welding and manufacture of steel.
(c) As oxygen cylinders in hospitals, high altitude flying and in mountaineering.
23
Answer
Ozone is an allotrope of ozone. It is formed from atmospheric oxygen in presence of sunlight.
Preparation:
When a slow dry stream of oxygen is passed through a silent electrical discharge in a special type of apparatus called ozoniser (Siemen’s and Brodie’s ozonisers) conversion of oxygen to ozone occurs. The product is known as ozonised oxygen :
3O2 → 2O3, ∆H°(298K) = +68kcal: 3O2 →2O3; ∆H° = +142kJ/mol
Properties
(a) It is a pale – blue gas with a characteristic smell. It changes to dark blue liquid and violet – black solid.
(b) It is harmless in small concentrations. In higher concentrations it can be explosive and can damage body tissues.
(c) It is heavier than air.
(d) O3 is neutral to litmus.
(e) It is thermodynamically unstable, it is endothermic i.e. its standard heat of formation is positive.
(f) It acts as a powerful oxidising agent due to the ease with which it liberates atoms nascent oxygen (O3 → O2 + O).
Some of the examples of its oxidizing action are :
- Pbs + 4O3 → PbSO4 + 4O2
- 2HCl + O3 → Cl2 + H2O + O2
- 2kl + H2O + O3 → 2KOH + I2 + O2
(g) Nitrogen oxides (emitted from jet aeroplanes) combine rapidly with ozone resulting in its depletion in upper atmosphere.
NO(g) + O3(g) → NO2(g) + O2(g)
Note :
(a) This reaction forms the basis of quantitative analysis of O3, O3oxidize I in a solution buffered with borate buffer having pH 9.2. The I2 formed is further reduced by a standard solution of Na2S2O3. The amount of Na2S2O3 gives the amount of I2 produced in oxidation which is then related to amount of O3 used in oxidizing I.
(b) Ozone layer in stratosphere protects earth from UV radiations but now it is under threat from nitrogenous oxides emitted from exhaust system of supersonic jets which are slowly depleting the concentration of ozone layer via reaction NO + O3 → NO2 + O2
Structure 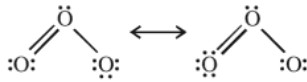
Uses :
(a) As germicide, disinfectant and for sterilising water.
(b) Bleaching oils, ivory, flour, starch, etc.
(c) Acts as an oxidising agent in the manufacture of KMnO4.
24
Answer
Important allotropes of sulphur are yellow rhombic (α – sulphur) and monoclinic (β – sulphur).
Yellow or rhombic sulphur (α – Sulphur)
- It is stable form of sulphur at room temperature.
- Its m.p. is 385.8K
- Its specific gravity is 2.06
- It is insoluble in water, somewhat soluble in benzene, alcohol, ether & readily soluble in CS2 .
Monoclinic sulphur (β – Sulphur)
- It is prepared by melting rhombic sulphur in a dish and cooling it.
- Its m.p. is 393 K
- Its specific gravity is 1.98
- It is soluble in CS2
- It is stable only above 369K
- Both α and β sulphur have S8 α – sulphur is stable below 369K and β – sulphur is stable above 369K and at 369K both the forms are stable.
- Other modifications of sulphur has 6 – 20 sulphur atoms per ring. In cyclo – S6 the ring adopts chair form.
- In vapour state at higher termperatures sulphur exists as S2 molecule which has two unpaired electrons in the antibonding π orbitals like O2 and hence, exhibits paramagnetism.
25
Answer
Preparation
(a) S(s) + O2(g) → SO2(g)
(b) In laboratory, by treating a sulphite with dil. H2SO4 .
SO32−(aq) + 2H+ (aq) → H2O (I) + SO2 (g)
(c) Industrially, as a by product of roasting of sulphide ores.
4FeS2(s) + 11O2(g) → 2Fe2O3(s) + 8SO2(g)
Properties
(a) Colourless gas with pungent smell and highly soluble in water.
(b) SO2 with water forms sulphurous acid. Thus it is acidic in nature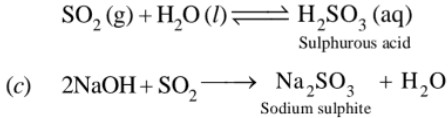
Sodium sulphite reacts with more SO2 to form sodium hydrogen sulphite.
Na2SO3 + H2O + SO2 → 2NaHSO3
(d) Reaction with chlorine in presence of charcoal as catalyst:
SO2(g) + Cl2(g) → SO2Cl2 (l)
(e) Sulphur dioxide reacts with oxygen in the presence of vanadium (V) oxide. ![]()
(f) In moist condition, acts as reducing agent.
For example :
2Fe3+ + 2H2O + SO2 → 2Fe2+ + SO42− + 4H+
It is an oxidising agent also,
Example of its oxidizing action are:
2H2S + SO2 → 2H2O + 3S
5SO2 + 2MnO4− + 2H2O → 5SO42− + 4H+ + 2Mn2+
Structure
It is angular and is a resonance hybrid of two canonical forms. 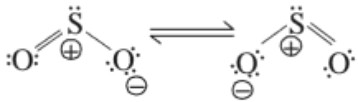
Uses:
(a) In refining petroleum and sugar.
(b) In bleaching wool and silk.
(c) As an anti – chlor, disinfectant and preservatives.
(d) In manufacture of NaHSO3, calcium hydrogen sulphite.
(e) As a solvent.
26
Answer
S, Se and Te forms a number of oxo – acids. Among the oxo – acids of S, sulphuric acid is most important. Other oxoacids of sulphur are H2SO3, H2S2O3, H2S2O4, H2S2O5, H2SxO6(x = 2 to 5) etc. Sulphurous acid (H2SO3) and thiosulfuric acid (H2S2O3) are unstable and cannot be isolated. They exist only in aqueous solutions or in the form of their salts.Structure of some important oxoacids of sulphur is as following:
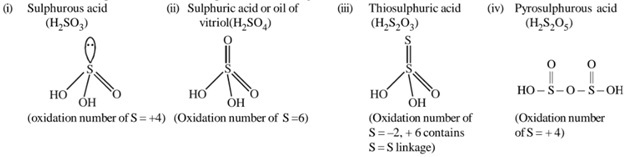
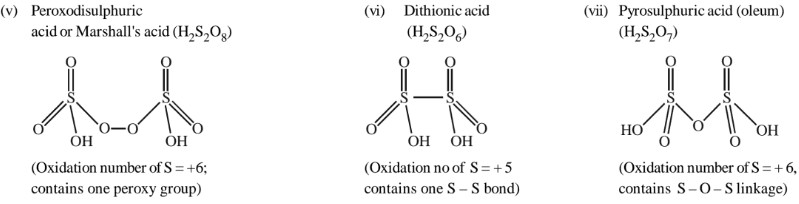
27
Answer
Preparation : Sulphuric acid is manufactured by contact process :
(a) Sulphur or sulphide ores burns in O2 to give SO2
S + O2 → SO2
(b) Catalytic oxidation of SO2 with O2 to give SO3 in the presence of V2O5. ![]()
(C) Then SO3 made to react with sulphuric acid of suitable normality to obtain a thick oily liquid called oleum.
SO3(g) + H2SO4(l) → H2S2O7(l)
Then oleum is diluted to obtain sulphuric acid of desired concentration.
H2S2O7(l) + H2O(l) → 2H2SO4(l)
Physical characteristic
(a) H2SO4 is colourless, dense, oily liquid.
(b) Freezing point and boiling point are 10.5°C and 340°C respectively.
(c) Density iss 1.84 gm cm−3.
(d) Forms hydrate with the evolution of heat :
H2SO4 . H2O [ monohydrate], H2SO4 . 2H2O [dehydrate],
H2SO4 .3H2O [trihydrate]
(e) It has strong affinity for water and acts as oxidizing agent.
(f) High b.p. and viscosity of H2SO4 is due to H–bondig . 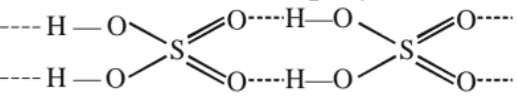
Chemical characteristics
(a) Acid character (dibasic acid)
H2SO4 → H+ + HSO4−, HSO4− → H+ + SO42−
(b) Forms two type of salts (normal and acidic):
NaOH + H2SO4 →NaHSO4 + H2O, NaHSO4 + NaOH → Na2SO4 + H2O
(c) Dehydrating agent [due to high affinity for water]![]()
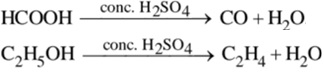
(d) Acts as an oxidizing agent
P4 + 10H2SO4 → P4O10 + 10SO2 + 10H2O, C + 2H2SO4 →CO2↑ + 2SO2↑ + 2H2O,
2HBr + H2SO4 → 2H2O + SO2↑ + Br2, H2S + H2SO4 → 2H2O + SO2↑ + S
(e) Displaces more volatile acids :
NaCl + H2SO4 → NaHSO4 + HCl, NaNO3 + H2SO4 → NaHSO4 + 2HNO3
FeS + H2SO4 → FeSO4 + H2S, Ca3(PO4)2 + 3H2SO4 → 3CaSO4 + 2H3PO4
(f) Reaction with metals : Zn, Mg, Fe gives hydrogen
Zn + H2SO4 (dil) → ZnSO4 + H2
Cu gives SO2.
Cu + 2H2SO4(conc.) → CuSO4 + 2H2O + SO2
Note : Metals like Cu, Pb, Hg, Bi and noble metals are not attacked by dil. H2SO4.
(g) Fromation of insoluble sulphates :
BaCl2 + H2SO4 → BaSO4↓ + 2HCl, Pb(NO3)2 + H2SO4 → PbSO4 ↓ + 2HNO3
(h) Reaction with PCl5 and KClO3 :
PCl5 + H2SO4 → ClSO2OH + POCl3 + HCl, 3KClO3 + 3H2SO4 → 3KHSO4 + HClO4 + 2ClO2 + H2O
Uses :
(a) Manufacture of fertilisers.
(b) Used in petroleum refining.
(c) Manufacture of pigments, paints and dyestuff intermediates.
(d) Used in detergent industry.
(e) Metallurgical aapplication.
(f) Used in storage batteries.
(g) Manufacture of nitrocellulose products.
(h) Laboratory reagent.
28
Answer
The group 17 of the periodic table contain fluorine (9F), chlorine (17Cl), bromine (35Br), iodine (53I) and astatine (55At). They are collectively called halogens as their salts are present in sea water. At is radioactive, artificially prepared and unstable. They have a strong tendency to accept one electron to acquire stable inert gas configuration. Hence exhibit non-metallic behaviour. High electronegativity makes them very reactive and they are not available in free state. Except At, the other members are found in combined state in suitable quantities in nature.29
Answer
(a) Electronic configuration : Electronic configuration is ns2 np5 for valence shells. 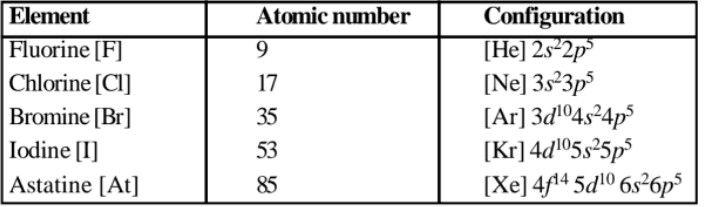
(b) Atomic and ionic radii : Halogens have the smallest atomic radii in their respective periods due to maximum effective nuclear charge. Down the group atomic and ionic radii increases from F to I due to increase in the number of quantum shells.
(c) Ionisation enthalpy : The first ionisation energies are relatively high out decrease down the group. Iodine can lose an electron and from I+ ion.
(d) Electron gain enthalpy : The electron affinities are high, increases from F to Cl and then decreases down the group. The electron affinity of F is less than that of Cl due to its small size with high electron density. Electron affinity varies as Cl > F > Br > I
(e) Electronegativity : Halogens have high electronegativities F is most electronegative element known. Electronegativity decreases on moving down the group.
(f) Physical state : Fluorine and chlorine are gases, bromine is a liquid while iodine is a solid.
(g)Melting and soiling points: Their melting and boiling points steadily increase with atomic number.
(h) Bond energy : Actually, the bond energy should have decreased from F – F to I – I but in practice it starts decreasing from Cl–Cl to I–I. ![]()
The lower value of bond dissociation energy of F – F bond Cl – Cl bond is due to larger inter – electron (electron – electron) repulsion between the non – bonding electrons in the 2p – orbitals of fluorine atom than those in the 3p – orbitals of chlorine atoms.
Moreover, the X – X bond in Cl2, Br2 and I2 is much stronger due to hybridisation of p – and d – orbitals.
(i) Non – metallic character : There is a gradual decline in the non – metallic character as we move down the group with decrease in electronegativity hence iodine shows some metallic character, viz. presence of metallic lustre and formation of positive ion, I+ .
(j) Density: It increases down the group in a regular fashion and follows the order F > Cl > Br > I.
30
Answer
(a) Oxidation state : All halogens exhibit – l oxidation state : Other halogens except fluorine also exhibit +1, +3, + 5 and +7 oxidation stae.
(b) Oxidising nature : Fluorine does not has d – orbital in its valence shell and hence cannot expand its octet. Therefore shows only –1 oxidation state. Halogens are good oxidising agents. The tendency of oxidising power decreases down the group . F2 is the strongest oxidizing agent and oxidizes other halide ions. A halogen oxidizes halide ions of higher atomic number.
(c) Reaction with metals and non – metals : X2 reacts with metals to give halides. The ionic character of the halides decreases in the order MF > MCl > MBr > MI
Halides of metals having low IE are more ionic in comparison to halides of metals having high IE. For metals exhibiting more than one oxidation states, the halide with higher oxidation state of metal is more covalent in comparison to halide having metal with lower oxidation state. X2 react with non – metals like S, P, As etc.
(d) Reaction with hydrogen : All halogens react with hydrogen to form volatile H – X, which dissolve in water to form hydrohalic acids. Acidic strength of hydrogen halides varies as :
HF < HCl < HBr < HI
The stability of these halides decreases down the group due to decrease in bond dissociation enthalpy in the order.
H–F > H–Cl > H–Br > H–I
Also volatility decreases from HCl to HI.
The order of b.p is HCl < Hbr < HI < HF
(e) Reaction with oxygen : Chlorine forms oxides Cl2O, ClO2 , Cl2O6 & Cl2O7.. These oxides are highly reactive and strong oxidising agents and tend to explode. ClO2 is used as bleaching agent for paper and pulp industries. It is also used in textiles and in water treatment.
Among the halogen oxides, bromine oxides Br2O2, BrO3 are least stable due to middle row anomaly. These are stable only at low temperature. These are powerful oxidants. Iodine forms I2O4, I2O5 and I2O7. These are insoluble solids. They decomposes in heating I2O5 is a good reducing agent. I2O5 is used in the estimation of CO.
(f) Reaction with metal : Halogens forms metal halides with metals. Ionic character of M – X bond decreases in the order M – F > M – Cl > M – Br > M – I
Halides in higher oxidation state are more covalent whereas halides in lower oxidation state are ionic
(g) Reaction with halogen : Halogens combine amongst themselves to form a number of compounds known as interhalogen of type XX’, XX3’, XX5’ and XX7
(h) Reaction with alkalies : Cl2, Br2 and I2 behave similarly when treated with alkali (they undergo disproportionation reaction)
- Cold and dilute alkali :
X2 + 2NaOH → NaX + NaOX + H2O - Hot and concentrated alkali :
3X2 + 6NaOH → 5NaX + NaXO3 + 3H2O
F2 behaves differently with alkalies :
F2 + 2NaOH(dil) → 2NaF + OF2 + H2O, 2F2 + 4NaOH(conc) → 4NaF + O2 + 2H2O
31
Answer
Anomalous behaviour of fluorine is due to its small size, highest electronegativity, non – availability of d – orbitals, low F – F bond dissociation energy and highest positive reduction potential.Main points of difference are :
(a) Fluorine shows only – 1 oxidation state. This is due to its most electronegative nature.
(b) Fluorine has a covalency of 1.
(c) Fluorine forms inter and intramolecular H – bonds.
(d) HF is a weak acid as compared of HCl, HBr and HI.
(e) Fluorine forms two series of salts NaHF2 and Na2F2.
(f) AgF is soluble in water while AgCl, AgBr and Agl are insoluble.
(g) Fluorine does not forms oxo acids while oxo acids of other halogens are well known, ex: HClO3, HOBr, HIO4 etc.
(h) Fluorine is the most reactive amongst halogens (due to low F – F energy).
(i) Fluorine forms SF6 whereas no other halogen form SX6.
(j) CaF2 is insoluble in water whereas CaCl2, CaBr2 and Cal2 are soluble.
32
Answer
Preparation :
(a) By heating MnO2 with conc. HCl.
MnO2 + 4HCl → MnCl2 + Cl2 + 2H2O
(b) By action of HCl or KMnO4.
2KMnO4 + 16HCl → 2KCl + 2MnCl2 + 8H2O + 5Cl2
Commercial manufacture
(a) In the Deacon process HCl is oxidised by air in presence of CuCl2 as catalyst at 400°C![]()
(b) In the electrolytic process, it is obtained at anode in the electrolysis of concentrated solution of brine (NaCl).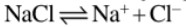
2Na+ + 2e− → 2Na + 2H2O → 2NaOH + H2 (at cathode); 2Cl− −2e− → 2Cl → Cl2 (at anode)
Properties
(a) Greenish – yellow gas with pungent and suffocating odour.
(b) Cl2 is 2 – 5 times heavier than air.
(c) Soluble in water its aqueous solution is known as chlorine water which on careful cooling gives chlorine hydrate Cl2.8H2O.
(d) Reacts with metals and non – metals to form chlorides.
For example : 2Fe + 3Cl2 → 2FeCl3 ; P4 + 6Cl2 → 4PCl3
(e) Chlorine has great affinity for hydrogen and react with hydrogen containing compounds to form HCl.
For e.g: H2S + Cl2 → 2HCl + S; H2 + Cl2 → 2HCl
(f) Reaction with NH3 : ![]()
(g) Reaction with NaOH ![]()
(h) Acts as oxidising and bleaching agent.
Oxidising action of Cl2
2FeSO4 + H2SO4 + Cl2 → Fe2 (SO4)3 + 2HCl
Bleaching action :
Bleaching action is due to oxidation
Cl2 + H2O → 2HCl + O, Coloured substance + O → Colourless substance
Uses :
(a) For bleaching pulp, cotton and textiles.
(b) Extraction of Au and Pt.
(c) Manufacture of dyes, drugs and organic compounds like CCl4, CHCl3, DDT, refrigerants, etc.
(d) Sterilising drinking water.
(e) Preparation of poisonous gases such as phosgene (COCl2), tear gas (CCl3NO2), mustard gas (ClCH2CH2SCH2CH2Cl).