The p-Block Elements
Book Solutions1
Answer
(i) Electronic configuration: There are 5 valence electrons for all the elements in group 15 (ns2 np3) is their general electronic configuration.(ii) Oxidation states: All these elements require three or more electrons to complete their octets and have 5 valence electrons. It is difficult in gaining electrons as the nucleus will have to attract three more electrons. This happens only with nitrogen as it is the smallest in size and the distance between the nucleus and the valence shell is relatively small. The remaining elements of this group show a formal oxidation state of −3 in their covalent compounds. In addition to the −3 state, N and P also show −1 and −2 oxidation states. All the elements present in this group show +3 and +5 oxidation states. However, the stability of +5 oxidation state decreases down a group, whereas the stability of +3 oxidation state increases. This happens because of the inert pair effect.
(iii) Ionization energy and electronegativity
Ionization decreases as we move down the group. This happens because of increase in atomic
sizes. Moving down the group, electronegativity decreases due to increase in size.
(iv) Atomic size: As we move down the group atomic size increases. This increase in
the atomic size is attributed to an increase in the number of shells.
2
Answer
Nitrogen is chemically less reactive. This is because of the high stability of its molecule, N2. In N2, the two nitrogen atoms form a triple bond. This triple bond has very high bond strength, which is very difficult to break. It is because of nitrogen’s small size that it is able to form3
Answer
4
Answer
When compared to phosphorus nitrogen is highly electronegative. This results in a greater attraction of electrons towards nitrogen in NH3 than towards phosphorus in PH3. Hence, the extent of hydrogen bonding in5
Answer
An aqueous solution of ammonium chloride is treated with sodium nitrite.
NH4Cl (aq ) + NaNO2 →N2(g) + 2H2O(l) + NaCl(aq)
NO and
6
Answer
Ammonia is prepared on a large-scale by the Haber’s process.N2(g) + 3H2(g) ⇌ 2NH3(g)
(i) Pressure (around
(ii) Temperature (4700 K)
(iii) Catalyst such as iron oxide with small amounts of
7
Answer
Concentrated nitric acid is a strong oxidizing agent. It is used for oxidizing most metals.
The products of oxidation depend on the temperature, concentration of the acid, and also
on the material undergoing oxidation.
3Cu + 8HNO3( dil.) → 3Cu(NO3)2 + 2NO + 4H2O
CU + 4HNO3( conc. ) →Cu(NO3)2 + 2NO2 +2H2O
8
Answer
9
Answer
Hydride NH3 PH3 AsH3 SbH3
H−M−H angle 107° 92° 91° 90°
The above trend in the H−M−H bond angle can be explained on the basis of the electronegativity of the central atom. Since nitrogen is highly electronegative, there is high electron density around nitrogen. This causes greater repulsion between the electron pairs around nitrogen, resulting in maximum bond angle. We know that electronegativitydecreases on moving down a group. Consequently, the repulsive interactions between the electron pairs decrease, thereby decreasing the H−M−H bond angle.
10
Answer
N (unlike P) lacks the d-orbital. This restricts nitrogen to expand its coordination number beyond four. Hence, R3N=O does not exist.
11
Answer
Nitrogen has a small size due to which the lone pair of electrons is concentrated in a small
region. This means that the charge density per unit volume is high. On moving down a
group, the size of the central atom increases and the charge gets distributed over a large
area decreasing the electron density. Hence, the electron donating capacity of group 15
element hydrides decreases on moving down the group.
12
Answer
Nitrogen owing to its small size has a tendency to form pπ−pπ multiple bonds with itself. Nitrogen thus forms a very stable diatomic molecule, N2. On moving down a group, the tendency to form pπ−pπ bonds decreases (because of the large size of heavier elements). Therefore, phosphorus (like other heavier metals) exists in the13
Answer
14
Answer
Catenation is much more common in phosphorous compounds than in nitrogencompounds. This is because of the relative weakness of the N−N single bond as compared
to the P−P single bond. Since nitrogen atom is smaller, there is greater repulsion of
electron density of two nitrogen atoms, thereby weakening the N−N single bond.
15
Answer
On heating, orthophosphorus acid (H3PO3) disproportionates to give orthophosphoric acid (H3PO4) and phosphine (PH3). The oxidation states of P in various species involved in the reaction are mentioned below.16
Answer
PCl5 can only act as an oxidizing agent. The highest oxidation state that P can show is +5. In PCl5, phosphorus is in its highest oxidation state (+5). However, it can decrease its oxidation state and act as an oxidizing agent.17
terms of electronic configuration, oxidation state and hydride formation.
Answer
The elements of group 16 are collectively called chalcogens.
(i) Elements of group 16 have six valence electrons each. The general electronic configuration of these elements is ns2np4, where n varies from 2 to 6.
(ii) Oxidation state:
As these elements have six valence electrons (ns2 np4), they should display an oxidation state of −2. However, only oxygen predominantly shows the oxidation state of −2 owing to its high electronegativity. It also exhibits the oxidation state of −1 (H2O2), zero (O2), and +2 (OF2). However, the stability of the −2 oxidation state decreases on moving down a group due to a decrease in the electronegativity of the elements. The heavier elements of the group show an oxidation state of +2, +4, and +6 due to the availability of d-orbitals.
(iii) Formation of hydrides:
These elements form hydrides of formula H2E, where E = O, S, Se, Te, Po. Oxygen and sulphur also form hydrides of type H2E2. These hydrides are quite volatile in nature.
18
Answer
Oxygen is smaller in size as compared to sulphur. Due to its smaller size, it can effectively form pπ−pπ bonds and form O2 (O=O) molecule. Also, the intermolecular forces in oxygen are weak van der Wall’s, which cause it to exist as gas. On the other hand, sulphur does not form M2 molecule but exists as a puckered structure held together by strong covalent bonds. Hence, it is a solid.19
Answer
More the lattice energy of a compound, more stable it will be. Stability of an ionic compound depends on its lattice energy. Lattice energy is directly proportional to the charge carried by an ion. When a metal combines with oxygen, the lattice energy of the oxide involving20
Answer
The aerosol which is responsible for the depletion of ozone is: Freons or chlorofluorocarbons (CFCs)
The molecules of CFS breaks down when there is presence of ultraviolet radiations and forms chlorine free radicals which then combines with ozone to form oxygen.
21
Answer
22
Answer
The environment is harmed by sulphur dioxide in many ways:
- Sulphuric acid is formed, when it is combined with water vapour present in the atmosphere. This causes acid that damages plants, soil, buildings (those made of marble are more prone) etc.
- SO2 causes irritation in respiratory tract, throat, eyes and can also affect the larynx to cause breathlessness.
- The color of the leaves of the plant gets faded when it is exposed to sulphur dioxide for a long time. This defect is known as chlorosis. The formation of chlorophyll is affected by the presence of sulphur dioxide.
23
Answer
Halogens have an electronic configuration of np5, where n =2-6. Thus, halogens require only one more electron to complete their octet and to attain the stable noble gas configuration. Moreover, halogens have high negative electron gain enthalpies and are highly electronegative with low dissociation energies. As a result, they have a high tendency to gain an electron. Hence, they act as strong oxidisong agents.24
Answer
Flourine has high electronegativity and a small size, hence it forms only one oxoacid i.e HOF.25
Answer
Oxygen has a smaller size and due to which a higher electron density per unit volume. Hence, oxygen forms hydrogen bonds while chlorine does not despite having the similar electronegative values.26
Answer
Applications of ClO2( a )Used for purification of water.
( b ) Used for bleaching.
27
Answer
Halogens arecolored because they take in radiations from the visible spectrum. This excites the valence electrons to a higher energy level. The amount of energy required for excitation differs from halogento halogen, thus they exhibit different colors.28
Answer
29
Answer
( i ) HCl is prepared from Cl2 by reacting it with water.
Cl2 + H2O → HCl + HOCl
( ii ) Cl2 is prepared by Deacon’s process from HCl
4HCl + O2 → 2Cl2 + 2H2O
31
( a ) H3PO3
( b ) PCl3
( c ) Ca3P2
( d ) Na3PO4
( e ) POF3
Answer
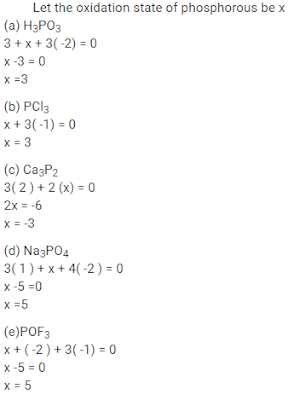
32
( a ) NaCl being heated with H2SO4 in the presence of MnO2.
( b ) Chlorine gas passed through aNaIand water solution.
Answer
(a) 4NaCl + MnO2 + 4H2SO4→ MnCl2 + 4NaHSO4 + 2H2O +Cl2
(b) Cl2 + NaI → 2NaCl + I2
33
Answer
XeF2, XeF4 and XeF6are obtained through direction reactions between Xe and F2. The product depends upon the conditions of the reaction :
Xe + F2→ XeF2
(excess)
Xe + 2F2→ XeF4
( 1:5 ratio )
Xe + F2XeOF4.
XeF2
(1 : 20 ratio)
34
Answer
ClO– is isoelectronic with ClF.Total electrons in ClO– = 17 + 8 + 1 =26
Total electrons in ClF = 17 + 9 = 26
As ClF accepts electrons from F to form ClF3, ClF behaves like a lewis base.
35
Answer
XeO3 can be obtained using two methods :
( 1 ) 6XeF4 + 12H2O → 4Xe + 2XeO3 + 24HF + 3O2
( 2 ) XeF6 + 3H2O → XeO3 + 6HF
XeOF4 is obtained using XeF6
XeF4 + H2O → XeOF4 + 2HF
36
( 1 ) Cl2, F2, I2, Br2 – increasing bond dissociation enthalpy.
( 2 ) HCl, HI, HBr, HF- increasing acidic strength.
( 3 ) PH3, NH3, AsH3, BiH3, SbH3 − increasing base strength.
Answer
(i) Bond dissociation energy usually decreases on moving down a group as the atomic size increases. However, the bond dissociation energy of F2 is lower than that of Cl2 and Br2. This is due to the small atomic size of fluorine. Thus, the increasing order for bond dissociation energy among halogens is as follows: I2 < F2 < Br2 < Cl2
(ii) HF < HCl < HBr < HI
The bond dissociation energy of H-X molecules where X = F, Cl, Br, I, decreases with an increase in the atomic size. Since H-I bond is the weakest, HI is the strongest acid.
(iii) BiH3 ≤ SbH3 < AsH3 < PH3 < NH3
On moving from nitrogen to bismuth, the size of the atom increases while the electron density on the atom decreases. Thus, the basic strength decreases.
37
(a) XeOF4
(b) NeF2
(c) XeF2
(d) XeF6
Answer
The one that does not exist is NeF2.38
( a ) ICl4–
( b ) IBr2–
( c ) BrO3–
Answer
39
Answer
Noble gases haveatomic radii that corresponds to van der Waal’s radii. Whereas, other elements have a covalent radii. Now, by definition van der Waal’s radii are bigger than covalent radii. This is the reason why noble gases have relatively bigger atomic sizes.40
Answer
Uses of Argon gas:
(a) Argon is used to keep an inert atmosphere in high temperature metallurgical operations like arc welding.
(b) It is used in fluorescent and incandescent lamps where it is required to check the sublimation of the filament. Thereby, increasing the life of the lamp.
(c) Argon is used in laboratories to handle substances that are air-sensitive.
Uses of neon gas:
(a) Neon is filled in discharge tubes for advertising or decoration.
(b) Neon is used for making beacon lights.
(c) It is used alongside helium to protect electrical equipments against high voltage.