NCERT Revision Notes for Polymers Class 12 Chemistry
CBSE NCERT Revision Notes1
Answer
Polymer which are obtained from animal and plants are known as natural polymers.
2
Answer
Individual molecule of very high molecular weight e.g., Heamoglobin, Chlorophyll etc.3
Answer
Natural, semisynthetic and synthetic compounds consisting of number of molecules (monomers) called Polymer. e.g, Polypropene, Polystyrene, PVC, Nylon – 6 etc.
Note :- The number of monomers which are joined together in a polymer constitutes the degree of polymerization. 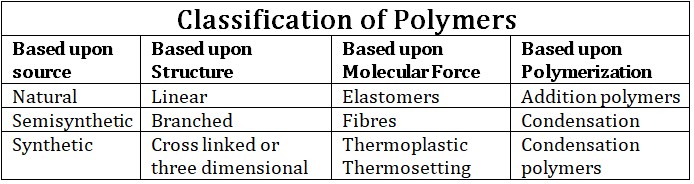
4
Answer
(i) Natural polymers :- Polymers which are found in nature E.g., Starch, Cellulose and natural rubber, Silk, Proteins, Nucleic acid….etc.
(ii) Semisynthetic polymers :- It is obtained from naturally occurring polymers by chemical treatment. Most of the semisynthetic polymers are prepared from cellulose e.g., cellulose acetate, cellulose nitrate, cellulose xanthate and rayon.
(iii) Synthetic polymers :- Man maid polymers are known as synthetic polymer e.g. PVC, polyethlene, polystyrene, nylon – 6 , nylon – 66; nylon – 6,10; terylene, synthetic rubbers etc.
Note:- Polymers in which repeating units are derived from two or more types of monomers units are called copolymers. Eg. Nylon – 6,6; Buna – S; Buna – N; Polyesters.
5
Answer
(i) Linear polymers :- Monomers are joined together to form long straight chains. The various linear polymeric chains are stacked over one another to give a well packed structure close packed in nature, having high densities, high melting point and high tensile (pulling) strength.Note :- All fibers are linear polymers. e.g. cellulose, silk nylon, terylene etc.
(ii) Branched chain polymers :- Polymers in which the monomeric units constitute a branched chain. Branched chain polymers have lower melting point low densities and tensile strength as compared to linear polymers. Examples are amylopectin, glycogen, low density polyethylene and all vulcanized rubbers.
(iii) Cross linked or Three :– Dimensional network polymers :- When linear polymeric chains are joined together to form a three dimensional network structure. These polymers are hard, rigid and brittle. Cross linked polymers are always condensation polymers. Resins are cross linked polymers.
6
Answer
(i) Elastomers :- Polymers in which the intermolecular foreces of attraction between the polymer chains are the weakest (weak van der Waals forces of attraction) these polymers consist of randomly coiled molecular chains of irregular shape having a few cross links. Examples are natural rubber, Buna – S, Buna – N etc.(ii) Fibres :- Those polymers in which the intermolecular forces of attraction are the strongest are called fibers. These polymers held together by H – bonding or dipole – dipole interaction. Fibres have high tensile strength, least elasticity having high melting point and low solubility.
(iii) Thermoplastics :- In thermoplastics intermolecular forces of attraction are in between those of elastomers and fibres. Thermoplastics become soft and viscous on heating and rigid on cooling. Examples are polythene, nylon – 6, nylon – 6, 6 etc.
(iv) Thermosetting polymers :- These polymers have low molecular masses and are semi – fluid substances. These polymers are hard and infusible. Examples are melamine – formaldehyde, bakelite( phenol – formaldehyde) etc.
7
Answer
1. Addition polymerization :-(a) Addition polymers are those in which addition reaction takes place
(b) If monomer is ethylene, then addition polymer may be either linear polymer of branched chain polymer. Examples are polystyrene, polytetrafluoroethylene, polyacrylonitrile etc.
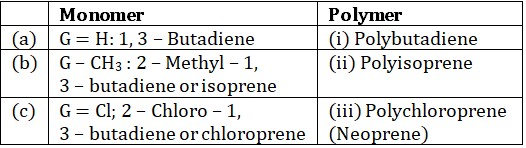
(c) If monomer is 1, 3 – butadiene or 2 – substituted – 1, 3 – butadiene
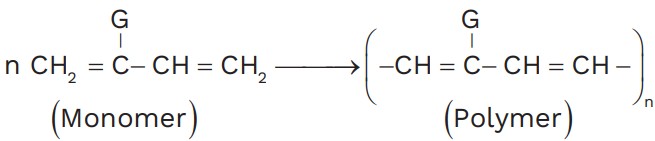
(d) Addition polymers are also known as chain growth polymers.
Types of Addition Polymerisation
(a) Free Radical Polymerisation
(i) Free radical polymerization takes in the presence of radical initiators. Such as dioxygen, benzoyl peroxide, acetyl peroxide etc.
For example, polymerization or ethane is carried out at high temperatures (350 – 570K) and at high pressure (1000 – 2000 atm) in presence of dioxygen or a small amount of benzoyl peroxide as radical initiator.
Mechanism
Chain initiating steps :
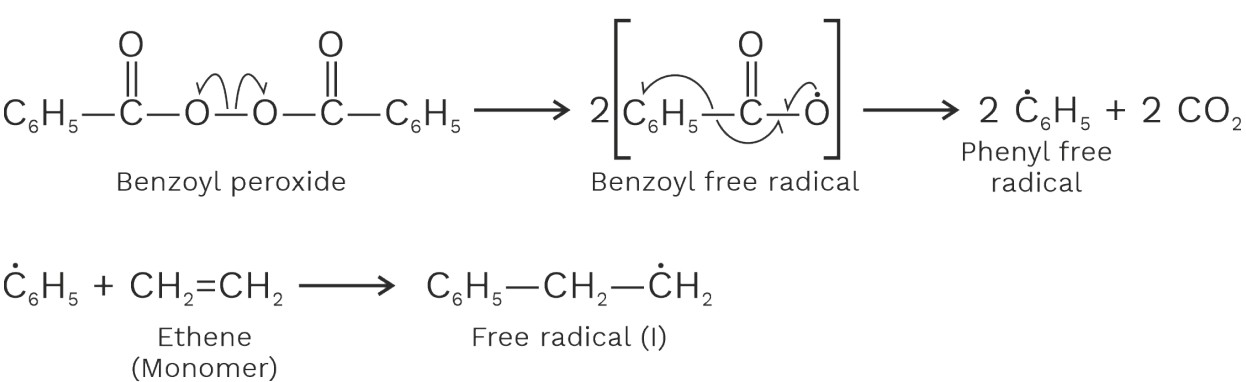
Chain propagating steps:
Chain terminating steps:
Step 1: By combination of free radicals (II)
Step 2 : By disproportionation of free radicals (II)
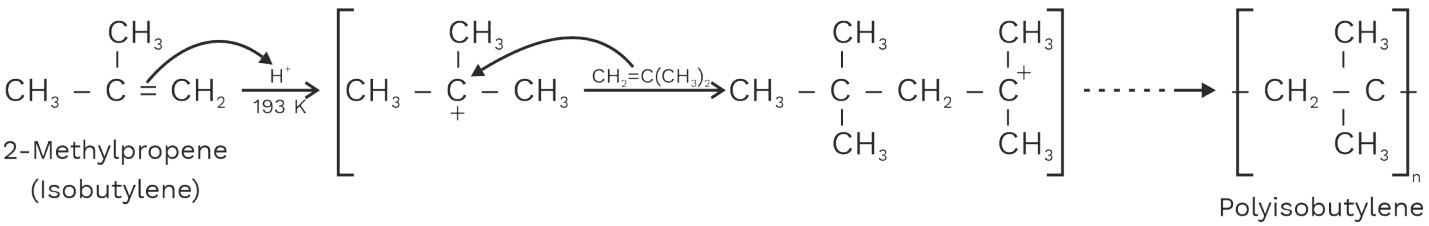
(b) Cationic Polymerisation
Cationic polymerization takes place in the presence of strong protonic acids such as H2SO4, AlCl3, BF3 etc.
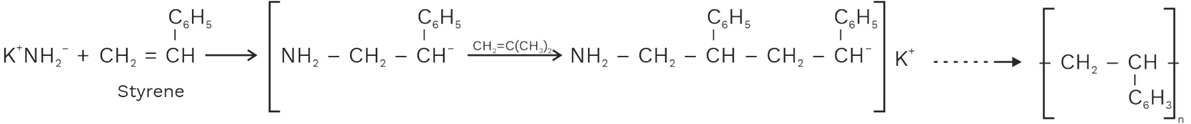
(c) Anionic Polymerization
In anionic polymerization, it takes place in the presence of strong bases such as KNH2, NaOH, KOH, some organometallic compounds etc.
2. Condensation polymerization
(a) They are formed due to condensation reactions.
(b) Condensation polymerization is also known as step growth polymerization.
(c) For condensation polymerization, monomers may should have at least two functional groups. (functional groups may be same or different).
For example,
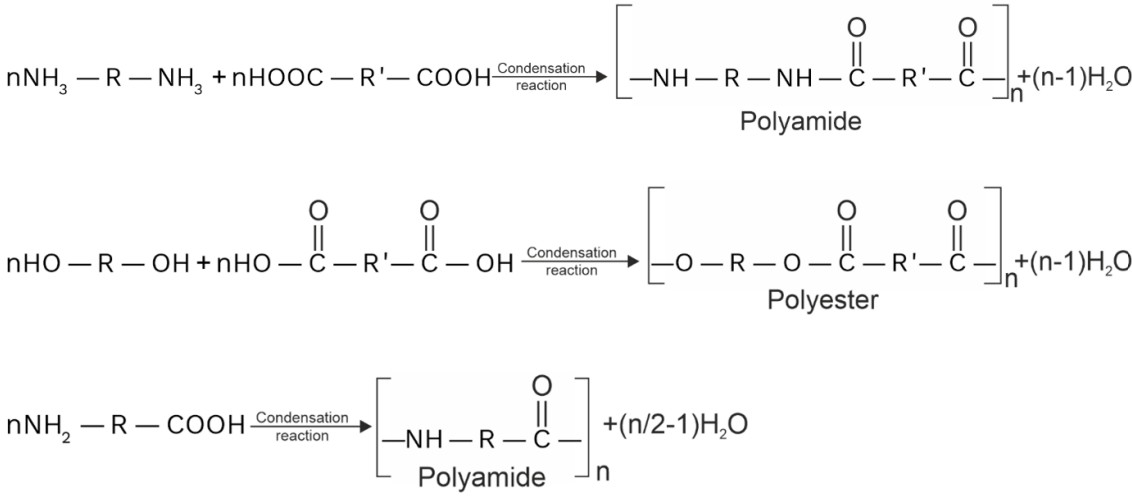
(d) During condensation elimination of small molecules like ammonia, alcohol, water, HCl takes places.
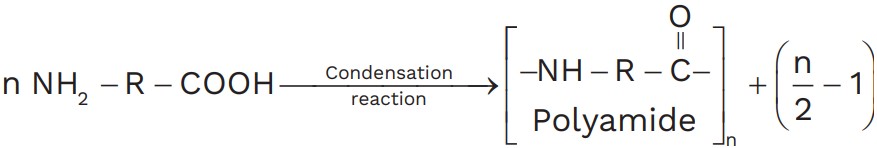
(e) Monomer having three functional groups always gives cross linked polymer. Examples are : Urea formaldehyde resin, phenol – formaldehyde resin.
8
Answer
The main source of natural rubber is braziliensis tree. Natural rubber is obtained from latex. Again latex is coagulated with acetic acid and formic acid and coagulated mass is then squeezed.Natural rubber is a sticky, gummy & soft and insoluble in water, alkalies &dilute acids. Natural rubber soluble in non – polar solvents. It has low elasticity and low tensile strength. Example : Polyisoprene.
Structure of Natural Rubber (cis – polyisoprene)
9
Answer
Polymers of 1, 3 – butadienes are called synthetic rubbers because they have some properties of natural rubber. Synthetic rubber are water proof and great elasticity.1. Homopolymers
Monomer of this class is 2 – substituted
Zieglar – Natta catalyst is used for polymerization which gives stereo regular polymers.
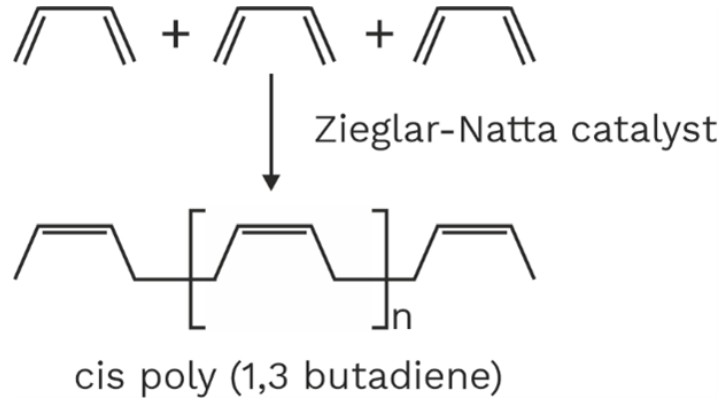
Dieprene (Neoprene) was the first synthetic rubber manufactured on large scale. It is the monomer of chloroprene (2 – chlorobutadiene).
2. Copolymers
Copolymers are derived from two or more types of monomer units. Example of copolymers are:
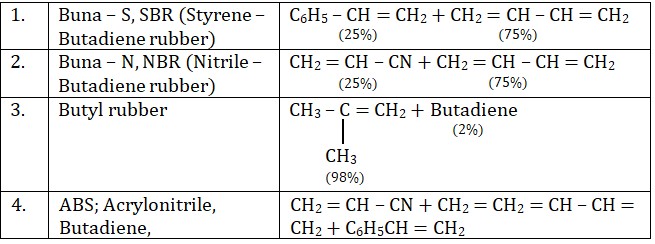
(a) Buna – S (SSR : Styrene – butadiene rubber)
Buna – S rubber is a copolymer or three moles of butadiene and one mole styrene. Buna – S is generally compounded with carbon black and vulcanized with sulpur. It is extremely resistant to tear & wear and therefore used in the manufacture of tyres and other mechanical rubber goods. It is obtained as a result of free radical copolymerization of its monomers.
(b) Buna – N
Buna – N is obtained by copolymerization of butadiene and acrylonitrile (General purpose Rubber acrylonitrile of GRA)
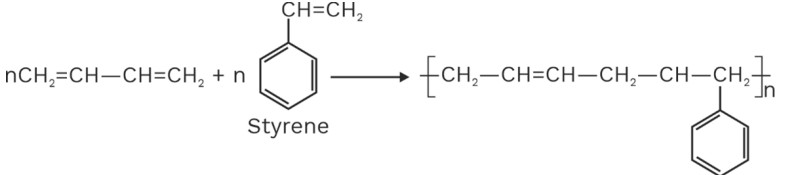
Buna – N is rigid and resistant to the action of organic solvents, lubricating oil and petrol. It is also used for making fuel tanks.
Vulcanisation of rubber
Natural rubber soft & sticky and becomes even more so at high temperatures and brittle at low temperanures. Therefore, rubber is generally used in the temperature range 283 – 335 K where its elasticity is maintained. It has a large water absorption capacity, has low tensile strength and low resistance to abrasion. It is also not – resistant to the action of organic solvents and is also easily attacked by axidising agents. These properties can be improved markedily by a process called vulcanization.
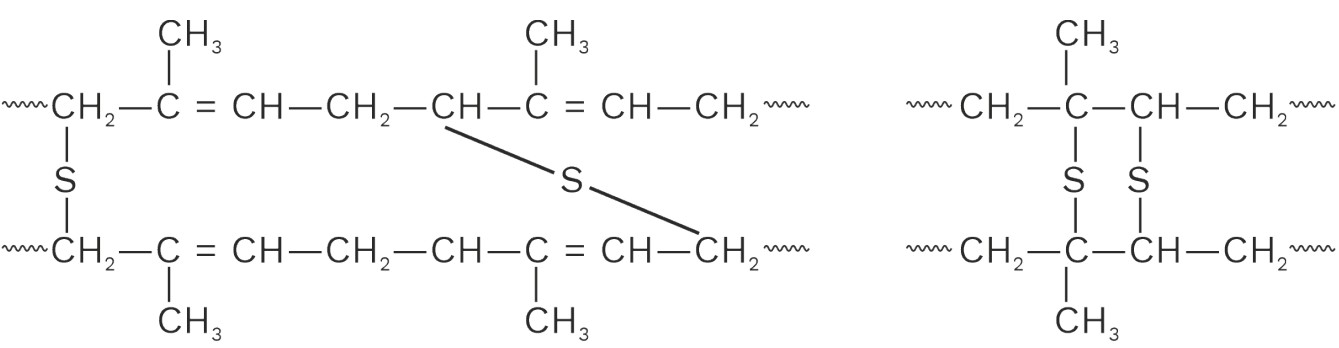
It consists of heating raw rubber with sulphur at 373 – 415 K. Since this process is slow, therefore, additives like zinc oxide, are used to acoelerate the rate of vulcanization the vulcanized rubber thus obtained has excellent elasticity, low water absorption tendency and is resistant to the action of organic solvents and oxidising agents During vulcanization, sulphur bridges or cross – links between polymer chans are introduced either reactive allylic positions or at the sites of the double bonds.
10
Answer
Nylons are prepared by the condensation polymerization of dibasic acids with diamines. Nylon contain amide linkages having a protein like structure.1. Nylon – 6, 6(Nylon – six, six)
It is obtained by condensation polymerization of a diamine with six carbon atoms (hexamethylenediamine) and a dibasic acid having 6 carbon atoms (adipic acid).
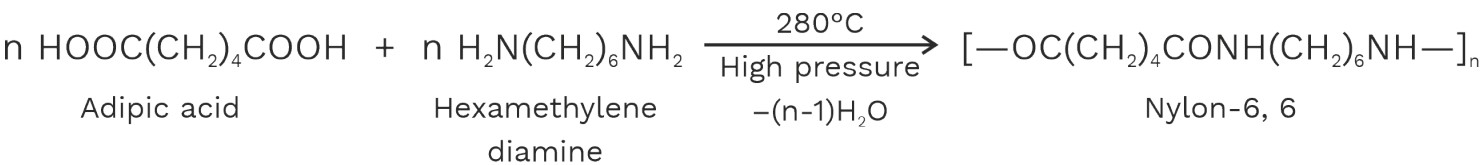
2. Nylon – 6, 10(Nylon – Six, ten)
It is obtained by condensation polymerization of six carbon atoms (hexamethylenediamine) and a dibasic acid with 10 carbon atoms (sebacic acid). These fibres are light, very strong, flexible and elastic having retain creases and drip dry property. These polymers are inert toward biological and chemicals agents. These polymers can be blended with wool to make carpets, garments, tyre cords, ropes, etc.
3. Nylon – 6 (Perlon L)
A polyamide closely related to nylon is known as perlon L (Germany) or Nylon – 6 (USA). During prolonged heating of caprolactum at 260 – 270°C. It is formed by self condensation of a large number of molecules of amino caproic acid. Since, caprolactum is more easily available, it is used for polymerization, with is carried out in the presence of H2O that first hydrolysis the lactam to amino acid. Subsequently, the amino acid can react with the lactam and the process goes on and onto formed by polyamide polymer.
Caprolactam is obtained by Backman rearrangement of cyclohexanone oxime.
11
Answer
Teflon is manufactured by heating tetrafluoroethene in presence of peroxides or ammonium persulphate catalyst at high pressures.12
Answer
During Polymerisation of acrylonitrile in the presence of a peroxide catalyst gives polyacrylonitrile.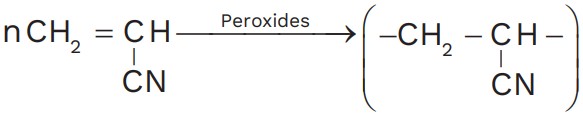
13
Answer
This resin is formed by condensation polymerization of melamine and formaldehyde. It is a quite hard polymer and is used widely for making plastic crockery under the name melamine. The articles made from this polymer do not break even when dropped from considerable height.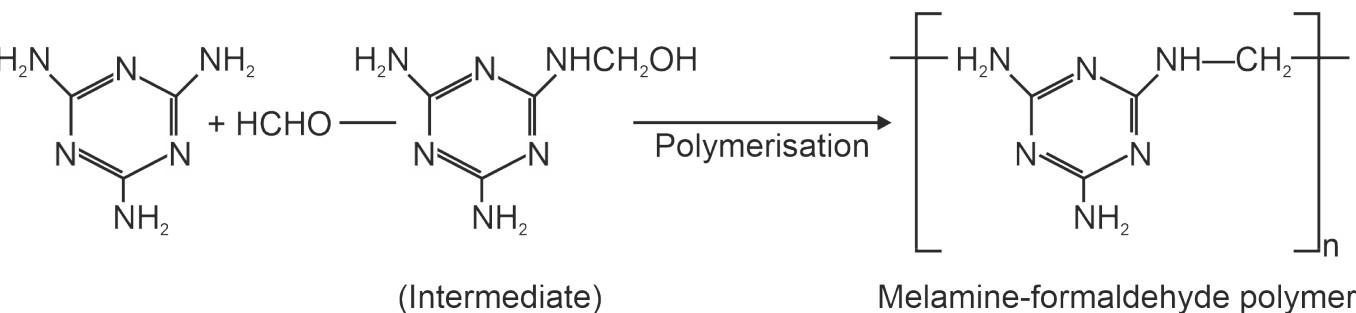
14
Answer
Phenol – formaldehyde resins are obtained by the reaction of phenol and formaldehyde in the presence of either an acid or a basic catalyst. It starts with the initial formation of ortho and para hydroxymethyl phenol derivatives, which further react with phenol to form compounds where rings are joined to each other with –CH2 groups. This reaction involves the formation methylene bridges in ortho, para or both ortho and para positions. Linear or cross – linked materials are obtained depending on the condition of the reaction.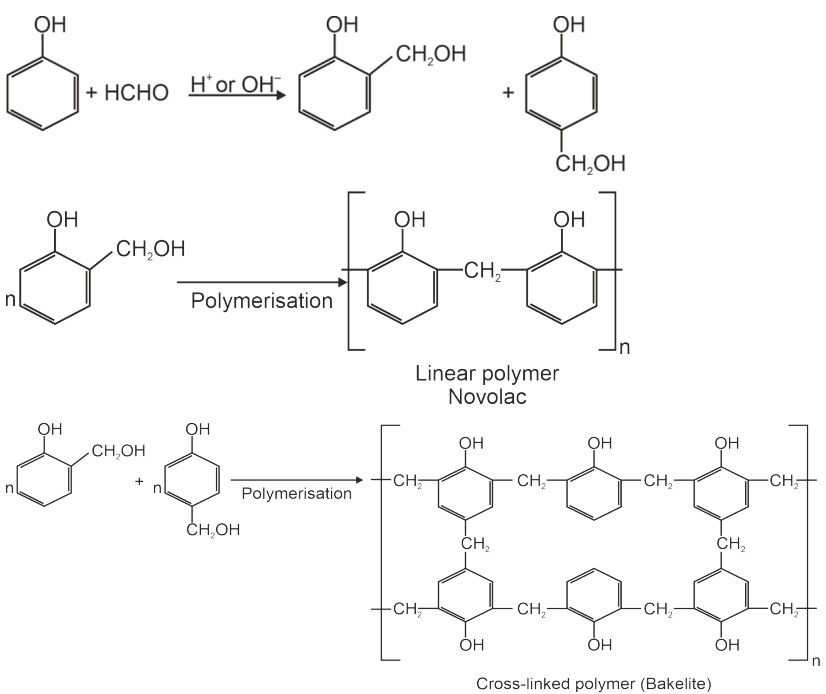
15
Answer
Dacron is a common polyester, prepared using ethylene glycol and terephthalic acid. The reaction is carried out at 140°C to 180°C in the presence of zinc acetate and Sb2O3 as catalyst.The Dacron is crease resistant and has low moisture absorption. It has high tensile strength. It is mainly used in making wash and wear gamets, in blending with wood to provide better crease and wrinkle resistance.
16
Answer
Normally, a polymer contains chains of varying lengths and therefore, its molecular mass is always expressed as an average. In contrast, natural polymers such as protein contains chain of identical length and hence, have definite molecular mass.Number average molecular mass
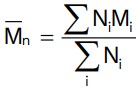
Here Ni is the number of molecules of molecular mass Mi .
Here Ni is the number of molecules of molecular mass Mi. Methods such as light scattering and ultracentrifuge depend on the mass of the individual molecules and yield weight as average molecular masses.
Some natural polymers, which are generally monodispersed, the PDI is unity (i.e.
In synthetic polymers, which are always polydisperse, PDI > 1 because
17
Answer
Many polymers are quite resistant to the environmental degradation processes and are thus responsible for the accumulation of polymeric solid waste materials (environmental pollution). These solid wastes cause acute environmental and health problems and remain undegraded for quite a long time (100 – 1000 years). For general awareness and concern, the problem created by the polymeric solid wastes, certain new biodegradable synthetic polymers have been designed and developed. These polymers contain functional groups similar to the functional groups present in biopolymers.By far the most important class of biodegradable polymers are aliphatic polyesters and polyamides. Some of these are discussed below :
(i) Nylon – 2 – Nylon – 6
It is an alternating polyamide of glycine (containing two carbon atoms) and ε – aminocaproic acid or 6 – aminohexanoic acid. (containing six carbon atoms)
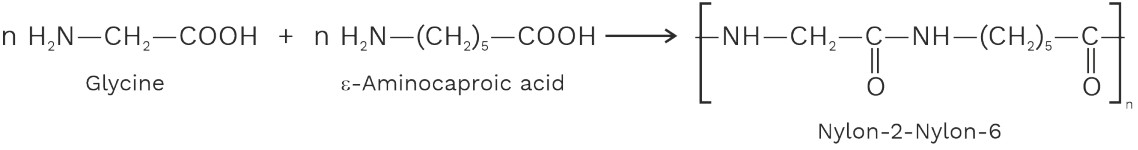
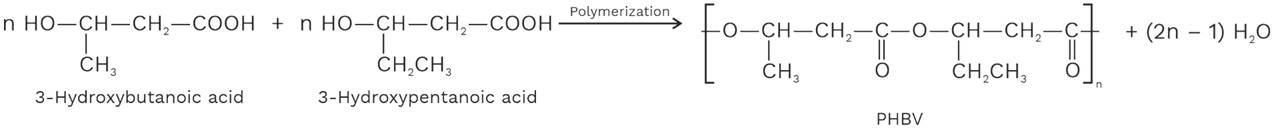
(ii) PHBV
It is a thermoplastic co – polymer of 3 – hydroxyl – butanoic acid and 3 – hydroxypentanoic acid in which the two monomer units are connected by ester linkages.