Polymers
Book Solutions1
Answer
Polymers are high molecular mass substances consisting of a very large number of simple repeating structural units joined together through covalent bonds in a regular fashion. Polymers are also called macromolecules. Some examples are polythene, nylon-66, bakelite, rubber, etc.
Monomers are the simple and reactive molecules from which the polymers are prepared either by addition or condensation polymerisation. Some examples are ethene, vinyl chloride, acrylonitrile, phenol and formaldehyde etc.
2
Answer
Natural polymers: Polymers which are found in nature, i.e., in animals and plants are called natural polymers, e.g., proteins, starch, cellulose, nucleic acids, resins and natural rubber.
Synthetic polymers: Man-made polymers are called synthetic polymers, e.g., plastics (polythene, PVC), synthetic fibres (polyester, nylon-6, 6) and synthetic rubber (neoprene, Buna-S).
3
Answer
Polymers whose repeating structural units are derived from only one type of monomer units are called homopolymers, e.g., PVC polyethene, PAN, teflon, polystyrene, nylon- 6 etc.
Polymers whose repeating structural units are derived from two or more types of monomer molecules are copolymers, e.g., Buna-S, Buna-N, nylon-66, polyester, bakelite.
4
Answer
Functionality means the number of binding sites in a molecule. For example, the functionality of ethene, propene, styrene, acrylonitrile is one while that of 1, 3-butadiene, adipic acid, terephthalic acid, hexa methylenediamine is two.
5
Answer
It is a process of formation of a high molecular mass polymer from one or more monomers by linking together a large number of repeating structural units through covalent bonds.
6
Answer
(−NH—CHR—C0−)n is a homo-polymer, the reason being that it is derived from a single monomer unit,7
Answer
On the basis of magnitude of intermolecular forces present in polymers, they are classified into the following groups:
(i) Elastomers
(ii) Fibres
(iii) Thermoplastic polymers
(iv) Thermosetting polymers
8
Answer
In addition polymerization, the molecules of the same or different monomers simply add on to one another leading to the formation of a macromolecules without elimination of small molecules like H2O, NH3 etc. Addition polymerization generally occurs among molecules containing double and triple bonds. For example, formation of polythene from ethene and neoprene from chloroprene, etc. In condensation polymerisation, two or more bifunctional trifunctional molecules undergo a series of independent condensation reactions usually with the elimination of simple molecules like water, alcohol, ammonia, carbon dioxide and hydrogen chloride to form a macromolecule. For example, nylon-6, 6 is a condensation polymer of hexamethylenediamine and adipic acid formed by elimination of water molecules.9
Answer
When two or more different monomers are allowed to polymerise together the product formed is called a copolymer, and the process in called copolymerisation. Example, Buna-S and Buna-N. Buna- S is a copolymer of 1, 3- butadiene and styrene while Buna-N is a copolymer of 1,3-butadiene and acrylonitrile.
10
Answer
(a) Chain initiation step: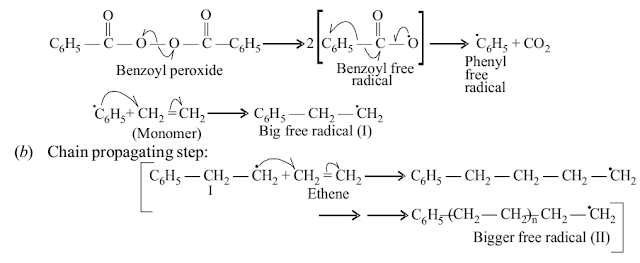

11
Answer
Thermoplastic polymers are linear (slightly branched) long chain polymers, which can be repeatedly softened and hardened on heating. Hence, they can be modified again and again. Examples include polythene, polystyrene.
Thermosetting polymers are cross-linked or heavily branched polymers which get hardened during the molding process. These plastics cannot be softened again on heating. Examples of thermosetting plastics include bakelite, urea-formaldehyde resins.
12
Write the monomers used for getting the following polymers.
(i) Polyvinyl chloride (ii) Teflon (iii) Bakelite
Answer
13
Answer
14
Answer
Natural rubber is cis-polyisoprene and is obtained by 1, 4-polymerization of isoprene units. In this polymer, double bonds are located between C2 and C3 of each isoprene unit. These cis-double bonds do not allow the polymer chains to come closer for effective interactions and hence intermolecular forces are quite weak. As a result, natural rubber i.e., cis–polyisoprene has a randomly coiled structure not the linear one and hence show elasticity.15
Answer
Natural rubber is useful but has problems associated with its use. The disadvantages of natural rubber are as follows:
1. Natural rubber is sticky and soft at room temperature. At elevated temperatures i.e grater than 335 K, it becomes even softer. At low temperatures i.e less than 283K, it becomes brittle. Therefore natural rubber can be used only at temperature range of 283 K-335 K to maintain its elasticity.
2. It absorbs large amount of water
3. It has low resistance to abrasion and low tensile strength.
4. It is soluble in non-polar solvents.
5. Can be easily attacked by oxidizing agents.
Vulcanization is done mainly to improve the properties of natural rubber. In this process, a mixture of raw rubber with sulphur and appropriate additive is heated at a temperature range between 373 K and 415 K.
16
Answer
The monomeric repeating
unit of nylon 6 is
The monomeric repeating unit of nylon 6, 6 is
17
(i) Buna-S (ii) Buna-N
(iii) Dacron (iv) Neoprene
Answer
18
Identify the monomer in the following polymeric structures:
Answer
19
Answer
20
What is a biodegradable polymer? Give an example of a biodegradable aliphatic polyester.
Answer