NCERT Revision Notes for General Principles and Processes of Isolation of Elements Class 12 Chemistry
CBSE NCERT Revision Notes1
Answer
There are 92 natural elements which constitute the entire matter on the earth. These are regarded as the building block of the universe. These are distributed in all three main parts of the earth, atmosphere and lithosphere.The naturally occurring metallic compounds found in earth crust which can be obtained by mining are called minerals.
Silicates and aluminates are the most abundant minerals but difficult to concentrate and reduce.
The d and p block metals are found as oxides and sulphides, except for group 3 metals which occur as phosphates. There is no mineral source of Tc (group 7) radioactive element that is made in nuclear reaction.
2
Answer
Metals occur in two forms in nature:-(1). Native (free or mineral) state
(2). Combined state
• Native ores : Ores contain metals in free state. Some of important metals are silver (Ag), gold(Au), platinum(Pt), mercury(Hg) etc.
•Sulphurised and arsenical ores : Such type of ores contains the sulphides or arsenides of metals:
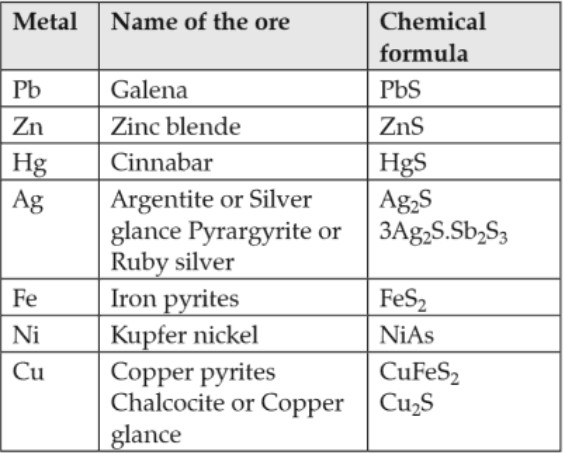
• Oxidised ores : Such types of ores contain oxides or oxy – salts such as carbonates, nitrates, sulphates, phosphates silicates etc.
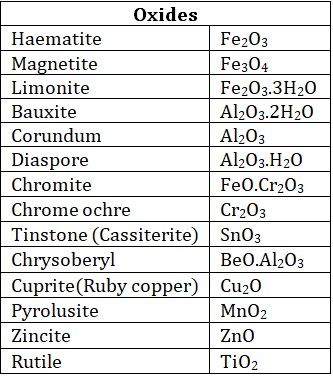
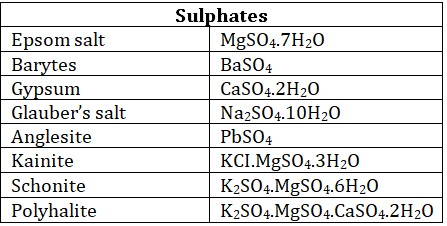
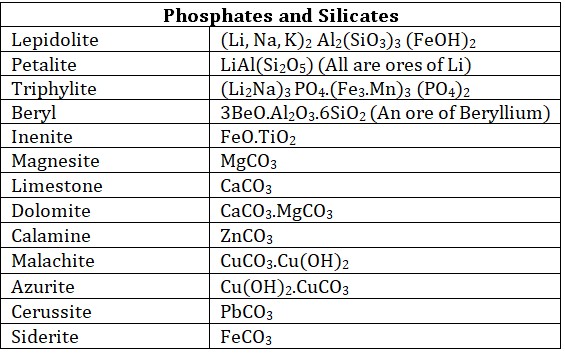
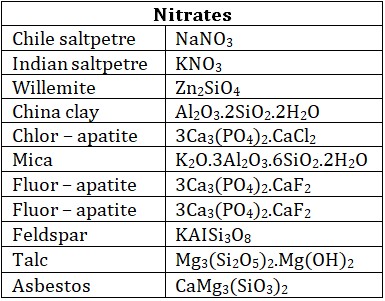
• Halide ores : Metallic halides are very few in nature, Chlorides are most common ores.
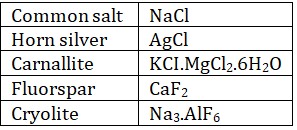
The minerals from which a metal can be conveniently, economically extracted in large quantity is known as ore.
[Note : “All ores are minerals but all the minerals are not ores’’]
Gangue : Ores are always contaminated with rocky and earthy impurities which is known as matrix or gangue. Silica is the common component of gangue.
3
Answer
Extraction of Al, Zn, Fe• Extraction of Al:-
1. Ores: (i) Bauxite : Al2O3.2H2O;
(ii) Cryolite : Na3.AlF6
2. Flow sheet of Baeyer’s process for the preparation of pure alumina :
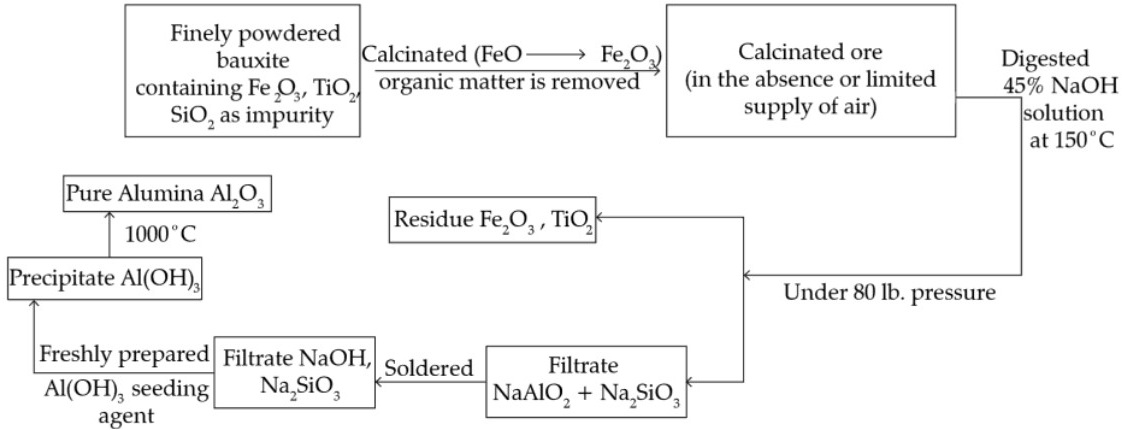
3. Electrolytic reduction :
Cathode : Iron tank lined with carbon bricks
Anode : Carbon
Electrolyte : Molten [Al2O3 20%, AlF3, 3NaF(60%), Fluorspar (CaF2)20%]
O2 is liberated at anode and AI collects at the bottom.
Reaction : Na3AlF6 → 3NaF + AIF3
AIF3 → Al3+ + 3F−
At anode : Al2O3 + 6F− → 2AIF3 + 3/2 O2 + 6e−
At cathode : 2Al3+ + 6e− → 2Al
4. Refining : Hoop’s process
Extraction of Zn :
(a) Ores :
(i) Zinc blende: ZnS
(ii) Calamine : ZnCO3
(iii) Zincite : ZnO
(b) Concentration :
(i) Froth flotation method
(ii) Roasting
2ZnS + 3O2 → 2ZnO + 2SO2
ZnS + 2O2 → ZnSO4
(c) Reduction : ZnO + C → Zn + CO
(d) Refining : Electrolytic refining, distillation.
Extraction of Fe :
(i) Ores :
Haematite : Fe2O3
Limosite : 2Fe2O3.3H2O
Magnetite : Fe3O4
(ii) Concentration : Electromagnetic separation.
(iii) Calcination and roasting : (Removal of H2O, S, C and As)
2Fe2O3.3H2O → 2Fe2O3 + 3H2O
FeCO3 →FeO + CO2
S + O2 → SO2
4AS + 3O2 →2As2O3
2Fe + 3/2 O2 → Fe2O3
Smelting (Blast furnace):-
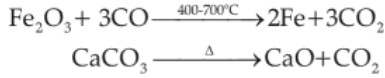
CaO + SiO2 → CaSiO3 (slag)
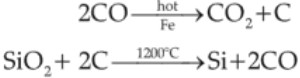
MnO2 + 2C → Mn + 2CO
P4O10 + 10C → P4 + 10CO
Pig iron : C = 3.1 – 4.5% small amount of Si, S, P, hard and brittle, obtained from blast furnace.
Wrought iron : C = 0.15 – 0.28%, purest form, malleable, fibrous.
Refining : By oxidising impurities in reverberatory furnace lined with haematite.
Fe2O3 + 3C → 2Fe + 3CO
4
Answer
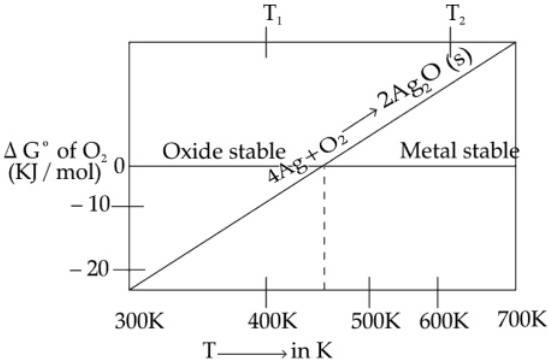
(i) Ellingham diagram is the variation of standard free energy change with temperature for the formation of oxides of various elements.(ii) These diagrams help as in predicting feasibility of thermal reduction of an ore.
(iii) ∆G = ∆H – T∆S, where ∆H, ∆S are constant with T unless a phase change takes place, hence ∆G vs T can be drawn as series of straight lines, where
∆S = slope, ∆H = intercept
The slope of the line changes when any of the materials involved melts or vaporizes.
(iv) The reaction for the formation of a metal oxide can be represented as
2x M(s) + O2(g) → 2MxOy (s)
⇒ xM(s) + ½O2(g) → MxO(s), (∆G°M, M2O)
∆G°T = ∆H°298 – T ∆ S°298
Therefore ∆ G°T vs T curves are straight lines.
∆G°T = A + BT where A = ∆H°298
(intercept) B = S°298k (slope)
y = c + mx
5
Answer
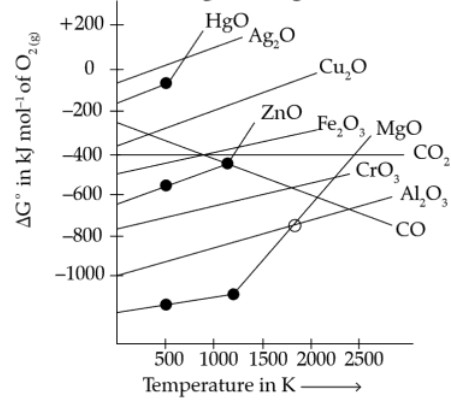
(i) Straight lines in the diagram (∆G° vs T) represents the relative stability of oxides.
(ii) From above graph we consider the oxides as
(a) Easily reducible oxides. e.g. : Au, Hg, Ag
(b) Oxides or greater stability . e.g. : FeO, MgO2
(c) Oxide which are very difficult to reduce : Al2O3, CaO, MgO.
(iii) ∆G° increases with increases of temperature as slopes of the curves for formation of metal oxide is positive.
(iv) Intercept of the curve represents the phase change.
(v) In Ellingham diagram, the plots of some reducing agents such as C, CO are also given the coupling or combination of oxidation and reduction helps in preceding whether the given reducing agent can be useful or not.
6
Answer
(i) It does not predict that, the kinetics of the reduction, i.e., rate or time taken for reduction to occur or how fast it could be.(ii) ∆G°does not take into account the activities of reactant or product which are other than unity.
7
Answer
(1) Collectors : Ethyl xanthate and potassium xanthate are used as collector in froth flotation process.
(2) Activator : It activates the ore and helps in separation of different minerals , present in some ores. e.g. CuSO4
(3) Depressant : The floating property of the component of the ore and helps to get separated from others by adding this e.g., NaCN, KCN are used as depressant.
(4) ![]()
(a) Acidic flux : SiO2
(b) Basic flux : CaO
(5) FeSiO3 is the slag formed in the extraction of copper.
(6) Cu2S + FeS is called matte.
(7) Bordeaux mixture: Solution of CuSO4 + Slaked lime
(8) Carnallite : Hydrated potassium and magnesium chloride KCl, MgCl2, 6H2O
(9) Chile saltpetre : NaNO3
(10) Ruby copper : Cu2O
(11) Calomel : Hg2Cl2
(12) Alnico : Alloy of Al containing :
Steel : 50% Al : 20% Ni : 10% C : 10%
(13) Alundum : Artificial alumina Al2O3
(14) Brimstone : S4/S8
(15) Cinnabar : HgS
(16) Coral : Naturally occurring calcium carbonate (CaCO3)
(17) Chalk Iceland spar, limestone, marble : CaCO3
(18) Carborundum : SiC
(19) Delta metal : Cu(55%) + Zn(40%) + Fe(10% or 15%) (alloy)
(20) Epsom salt : MgSO4.7H2O
(21) Fusion mixture : Na2CO3 + K2CO3 ![]()
(23) Glauber salt : Na2SO4.10H2O
(24) Graham’s salt : Calgon (NaPO3)6 or Na2[Na4(PO3)6]
(25) Gun powder : KNO3 + S + Charcoal
(26) Horn silver : AgCl
(27) Lithopone : ZnS + BaSO4
(28) Microcosmic salt : Na(NH4)HPO4.4H2O
(29) Mosaic gold : SnS2
(30) Rouge : Fe2O3
(31) Milk of magnesia : Mg(OH)2
(32) Pearl ash : K2CO3
(33) Purple of Cassius : AuCl3 + SnCl2
(34) Philosopher’s wool : ZnO
(35) Quick lime : CaO
(36) Quicksilver : Hg
(37) Quartz : SiO2
(38) Shora : KNO3
(39) Super phosphate of lime : Ca(H2PO4)2.H2O
(40) Silica gel : SiO2. xH2O
(41) Salt cake : Na2SO4
(42) Inorganic sugar : Pb(CH3COO)2.3H2O
(43) Minium : Pb3O4 (Red lead)
(44) Talc : Hydrated oxide of Mg and Sr.
(45) Fool’s Gold : FeS2
(46) Britannia metal : Sn 93%, Sn 5%, Cu2%,
(47) Rolled Gold : Cu80%, Al 10 – 20%.
(48) Duralumin : Al 95%, Cu 4%, Mn 0.5%, Mg 0.5%
(49) The process of extraction of Au by leaching is known as Mac Arthur Forest Cyanide Process.
4Au + 8KCN + 2H2O + O2 → 4K[Au(CN)2] + 4KOH
2K[Au(CN)2] + Zn →K2[Zn(CN)4] + 2Au↓
(50) Steel : 0.5 – 1.5% of C + with small amounts of other elements [Mn, Cr, Ni etc.] with Fe.
(51) Fe2O3.H2O → Iron oxide yellow
(52) German silver is an alloy of Cu(60%), Zn(20%), Ni(20%).
(53) ZnS is used for preparing luminous dials for watches and X – ray screens.
(54) CuSO4.5H2O → Blue vitriol/Nila thotha
(55) Cu (CH3COO)2, Cu(OH)2 : Basic copper acetate verdigris.
(56) Schweizer’s reagent : [Cu(NH3)4 (H2O)2] (OH)2
(57) The process of coating iron sheet with Zn → Galvanisation
(58) Molten Ag dissolves 20 times its volume of O2 which is given off on cooling causing violent spurting : splitting of silver.