NCERT Revision Notes for Chapter 5 Coordination Compounds Class 12 Chemistry
CBSE NCERT Revision Notes1
Answer
When two or more than two simple salts are allowed to chemically combine in a fix ratio then addition compound is formed. On the basis of behaviour in aqueous solution they are of the following two types.
1. Double salt: - Addition compound in which simple salts don’t loss their identity and its aqueous solution give test of its all constitute ions.
• Double salts loss their identity in aqueous solution.
KCl + MgCl2 + 6H2O → KCl. 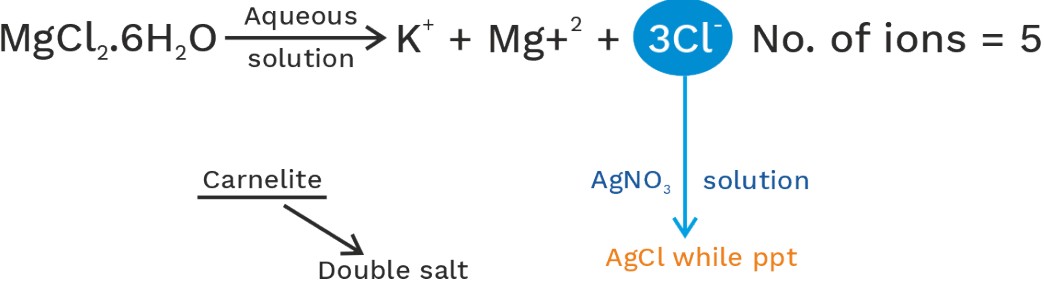
• All Alums are double salt.
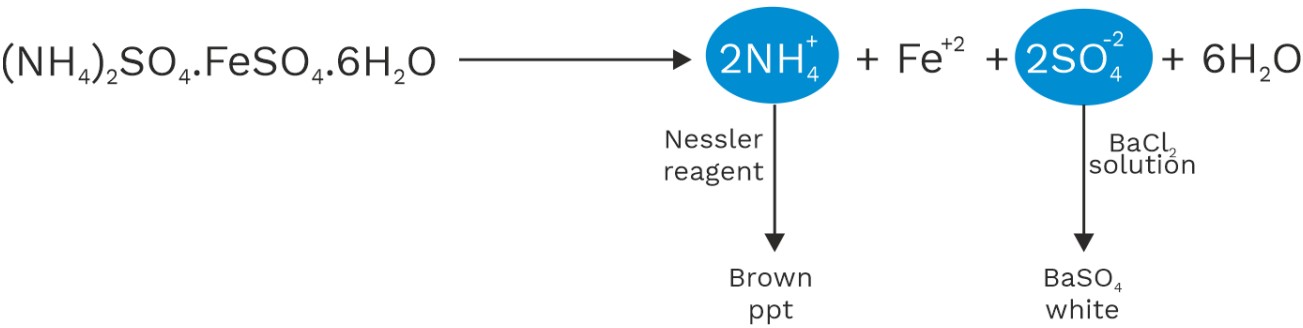
2. Complex compound : − Addition compound in which simple salts loss their identity and it’s aqueous solution doesn’t give test of its all constitute ions.
• Complex compound don’t loss their identify in aqueous solution.
K4[Fe(CN)6] → 4K+ + [Fe(CN)6]4−
Does not ionize to give Fe2+ and CN− ions
2
Answer
1. On basis of type of ligands(i) Homoleptic complex: - Same type of ligands
Ex.
(ii) Heteropoietic Complex:- Different type of ligands

2. On the basis of degree of dissociation
(i) Perfect complex compound:- Relatively more stable and its aqueous solution doesn’t give test of its all constitutive ions.
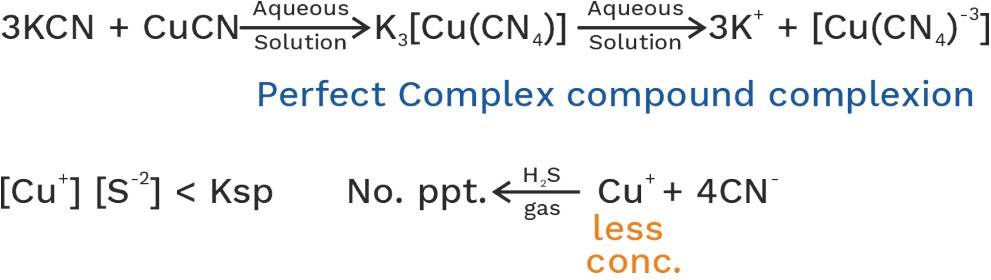
(ii) Imperfect complex compound: - Relatively less stable and its aqueous solution gives test of its all constitute ions.
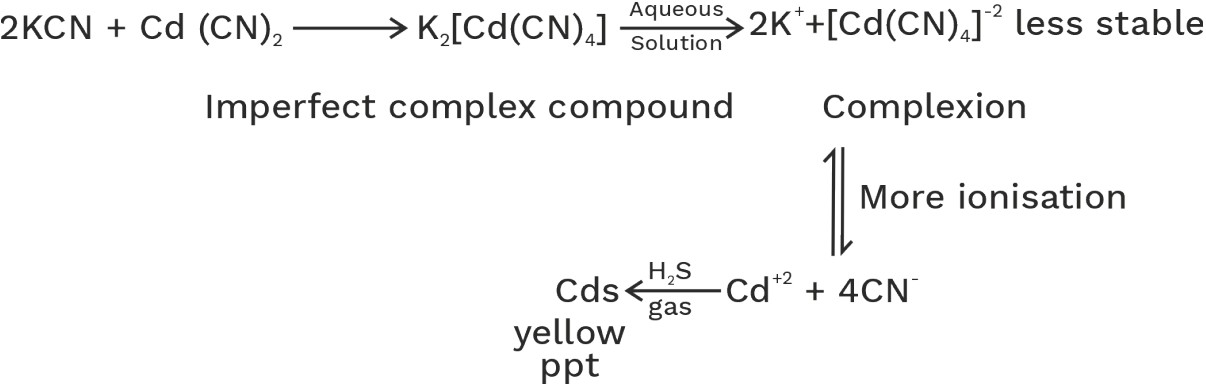
• Imperfect complex compound which undergoes 100% ionisation is known as double salt.
• There is no sharp line between perfect and imperfect complex compound.
• Complex forming tendency decreases down a group in s-block because size increases.
3
Answer
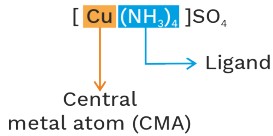
Coordination sphere :- ![]()
Outside region:-
• Ionisation sphere
• Free ion
• Cation
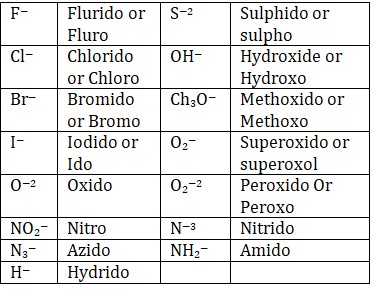
Naming of ligands
• —O—suffix provided to the name of anionic ligands.
• —ium suffix provided to the name of cationic ligands.
Anionic ligands ending with -ide are named by replacing -ide with suffix -o or replacing -e by -o. Ligands whose names end in -ite or -ate become -ito or -ato, i.e., by replacing the ending -e with -o.
4
Answer
(a) On the basis of denticity
1. Monodentate ligand
(i) Neutral ligands 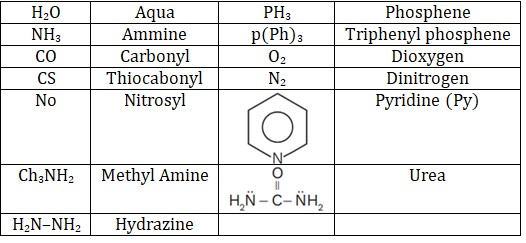
• Hydrozine never acts as bidentate ligand.
(ii) Cationic ligand : - 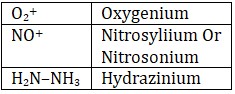
(iii) Anionic ligands
2. Bidentate ligands
(i) Ethylene diamine (en) or (ethane 1, 2−diamine)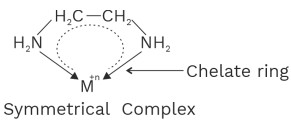
(ii) Propane 1, 2–diamine Or propylene diamine (Pn) 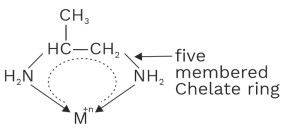
(iii) Dipyridyl (dipy)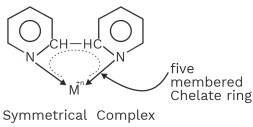
(iv) Oxalato (OX)−2 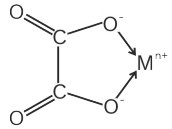
(v) Glycenato (gly)− 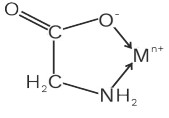
(vi) Dimethyl glyoximato (dmg)− 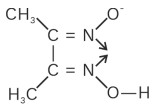
3 Polydentate ligands
All bidentate and polydentate ligand are chelating ligands.
No of chelate ring = Denticity – 1
(i) Diethylenetriamine (dien)
Denticity = 3
No. of rings = 2
Neutral 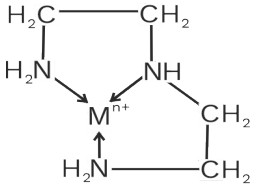
(ii) (trien) – Triethylene tetramine
Denticity = 4
Chelate rings = 3
Neutral
(iii) Terpyridine
Denticity = 3
Chelate rings = 2
Neutral
(iv) Ethylenediamine tetracetato (EDTA)–4
2N 4(O)
Denticity = 6
Chelate rings = 5 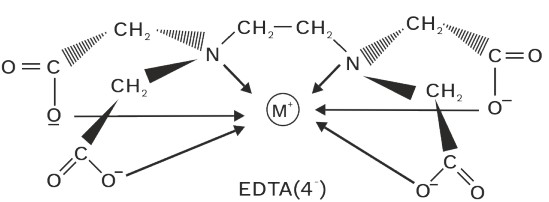
(v) Ethylenediamine triacetate (EDTA)–3
Denticity = 5
Chelate rings = 4
4 Ambidentate ligands
• Ligands which have two different donor atoms but at time of coordination such ligand can be coordinate CMA by either of two donor atoms. 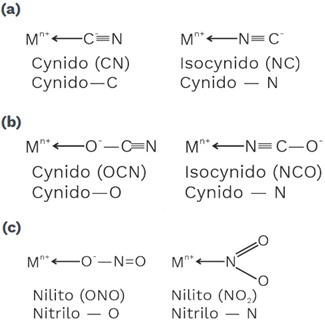
5 Flexi dentate
(a) Ligands which can change their denticity
Ex. CO3−2, PO4−3, SO4−2, CH3 – COO−, NO3− etc. 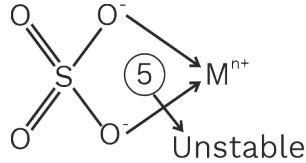
Ex. [Co(NH3)5 SO4]Cl
x + 0 – 2 = 0
x ⇒ +3
C.N ⇒ 6
5 by NH3 + 1 by SO4
Monodentate
(b) On the basis of e− donating and e− accepting nature
(i) Normal Or classical ligands
Ligands which only donate e− pair to CMA and form coordinate σ bond.
Ex. NH2−, OH−, N−3, Cl− , O–2
(ii) Non classical or π acid or π acceptor ligands
Ligands which donate e- pair to CMA and form coordinate bond but simultaneously they accept e- pair from CN through back bonding (synergic bonding)
Ex. CO,NO+ ,CN− ,C6H6,C5H5−, C2H4
(iii) π donor and π acceptor ligand
Ligands which donate π e− to CMA and also accept e− density from CMA though synergic bonding.
Ex. C6H6, C5H5−
(a) 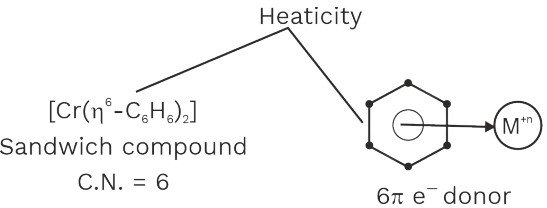
(b) 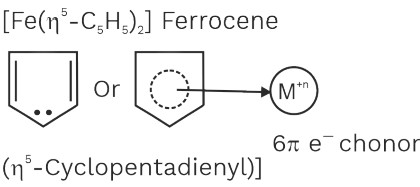
5
Answer
Total no of e− pair accepted by CMA
1. [Ni(dmg)2] = 4
2. [Pt(trien)]Cl2 = 4
3. [Fe(EDTA)]− = 6
4. [Co(en)2Ox]Cl = 6
Some important points
(i). Generally C.N of monovalent cation is two and four except
Ex. 
(ii) Generally C.N or bivalent cation is four and six except. ![]()
(iii) Generally C.N of trivalent cation is six except some exceptions.
(iv) C.N of tetravalent cation is 6.
(v) C.N of CMA depends upon charge of CMA, six of CMA size of ligands and concentration of ligand.
6
Answer
Total number of e− of CMA after accepting e− pair from ligands
ENA = Z – (0.5) + 2 × C.N.
1. K4[Fe(CN)6] ⇒ 26 – (+2) + 2 × 6 ⇒ 36[K]r
O.S. = +2, C.N. = 6
2. K3[Fe(CN)6] ⇒ 26 – (+3) + 2 × 6 ⇒ 35[Kr]
O.S. = + 3, C.N. = 6
3. [Fe(η5 – C5H5)2] ⇒ 26 – (+2) + 2× 6 ⇒ 36[Kr]
O.S. = +2, C.N.= 6
Sidgwick rule
• If EAN of CMA in metal carbonyl is equal to Atomic number of nearest inert gas then the stability of metal is high. 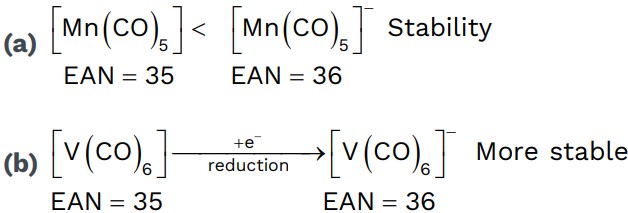
(C) [Fe(CO)5] ⇒ neither oxidising nor reducing
• Sidgwick rule is applicable only for metal carbonyl.
EAN of Polynuclear Metal Carbonyl 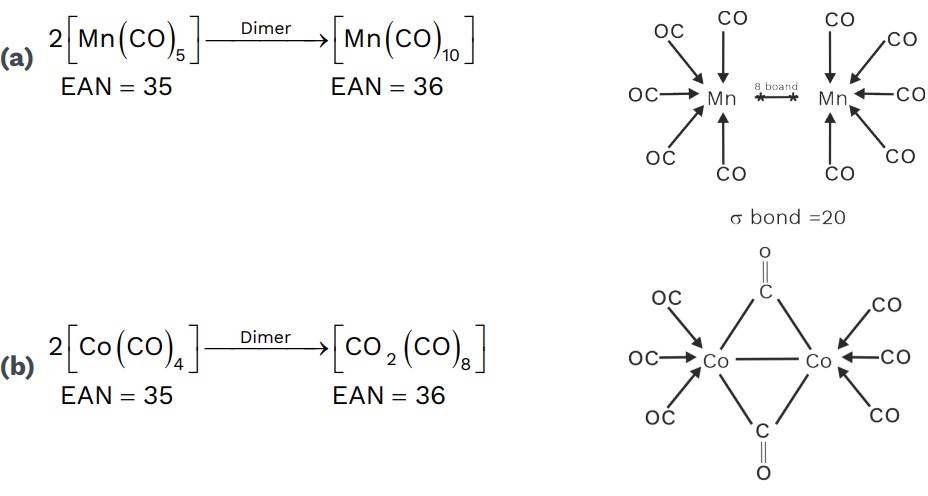
7
Answer
1. Formula of a Complex
(a) In formulas of both simple and complex salts, cation precedes the anion. Nonionic compounds are written as single units.
(b) Complex ions are written inside square brackets without any space between the ions.
(c) Metal atom and ligands are written in the following order:
- In the complex part, the metal atom is written first followed by ligands in the order, anionic → neutral → cationic.
- If more than one ligand of one type (anionic, neutral or cationic) are present, then they are arranged in English alphabetical order, e.g. between H2O and NH2, H2O should be written first. Similarly, order of NO2− , SO32− and OH– will be NO2−
- When ligands of the same type have similar name for the first atom, then the ligand with less number of such atoms is written first. Sometimes the second atom may be used to decide the order. When number of atoms are also same e.g., Out of NO2− , NH2− will be written first. In H3 and
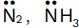 will be written first as it contains only one N-atom.
will be written first as it contains only one N-atom.
- Polyatomic ligands and abbreviations for ligands are always written in lower case letters. e.g. (en), (py), etc.
- Charge of a complex ion is represented as over script or square bracket.
Examples:
K4[Fe(CN)6] — First cation and then anion
[CrCl2(H2O)4] Br—Cl– (negative ligand) before H2O
2. Nomenclature of Coordination Compounds
Mononuclear coordination compounds are named by following these rules:
(a) In both the positively and negatively charged coordination compounds, the cation is named first followed by the anion.
(b) The name of the central atom/ion is written after the ligands are named in alphabetical order. (This procedure is reversed in writing its formula).
(c) Names of the anionic ligands end in –o.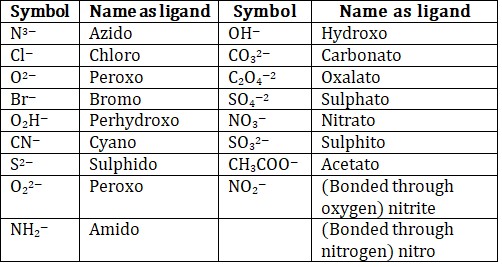
(d) Names of neutral and cationic ligands are the same except for aqua for H2O, ammine for NH3, carbonyl for CO and nitrosyl for NO. These are placed within parentheses (). 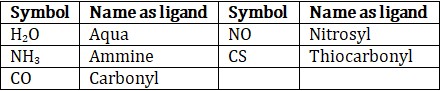
(e) Positive ligands are named as : 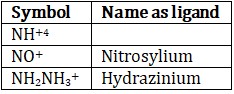
(f) Oxidation state of the metal in a cation, anion or a neutral coordination compound is indicated by a Roman numeral in parenthesis.
(g) When the complex ion is a cation, the metal is named same as the element. For example, Co in a complex cation is called cobalt and Pt is called platinum. In an anion, Co is called cobaltate. For some metals, their Latin names are used in the complex anions, e.g. ferrate for Fe.
(h) Nomenclature of a neutral complex molecule is done in the similar way as that of a complex cation.
The following examples illustrate the nomenclature for coordination compounds:
• [Cr(NH3)3(H2O)3] Cl3 is named as: Triamminetriaquachromium (III) chloride
• [Co(H2NCH2CH2NH2)3]2 SO4 is named as: Tris (ethane –1, 2–diammine) cobalt (III) sulphate
• [Ag(NH3)2] [Ag(CN)2 ] is named as: Diamminesilver (I) dicyanoargentate (I)
(i) Ligands which join two metals are known as ‘Bridge ligands’ and they are prefixed by ‘µ’ (mu).
E.g. 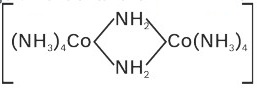 in this complex.
in this complex.
3. Nomenclature of Complexes
(a) Cationic Complex
[Cr(NH3)3(H2O)3]Cl2
triamminetriaquachromium (III) chloride
(i) The number of the individual ligands are indicated by prefix like mono, di, tri, etc. and ligands are named in an alphabetical order.
(ii) Central metal atom and oxidation state indicated by Roman numeral in parenthesis.
(iii) Name of ionisable anion.
(b)Anionic Complex
K3[Fe(CN)6]
Potassium hexacyanoferrate (III)
(i) Name of ionisable metal and oxidation state
(ii) Name of ligand in an alphabetical order
(iii) Central metal atom + ate and oxidation state
(c) Neutral Complex
[Pt(NH3)2Cl(NO2)]
Diammine chloronitrito–N–platinum (II)
(i) Name of ligands in an alphabetical order
(ii) Central metal atom and oxidation states.
8
Answer
Ferromagnetic : Fe, Co, NO2Spin magnetic moment = (μ) =
n = 1 μ = 1.73
n = 2 μ = 2.83
n = 3 μ = 3.87
n = 4 μ = 4.90
n = 5 μ = 5.92
Spin magnetic moment of bivalent cation of 3d series
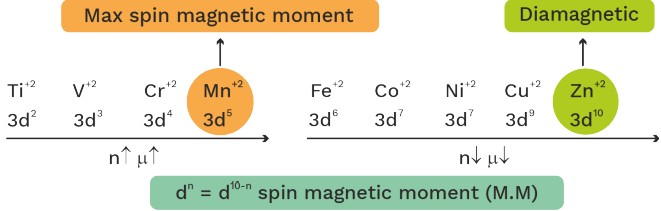
Spin magnetic moment of trivalent lanthanoids
• ‘Na’ (Neodymium) has max. M.M. among lanthanoids due to sum of spin magnetic moment + orbital magnetic moment.
9
Answer
1. Werner’s TheoryAccording to Werner, central metal atom has two types of valencies
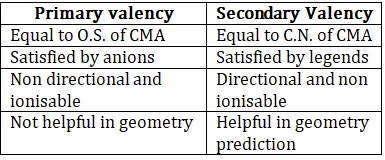
• All secondary valencies have specific arrangement according to C.N.
CN = 2 → Linear
CN = 3 → Trigonal planar
CN = 4 → Tetrahedral or square planar
CN = 5 → TBP or square pyramidal
CN = 6 → Octahedral
[Co(H2O)6]Cl3
O.S. ⇒ +3CN = 6
Primary valency = +3
Secondary valency = 6
Satisfied by 3Cl Satisfied by 6H2O
• Possible complex of CaCl3. 6H2O
O.S = +3 C.N. = 6
(a) [Ca(H2O)6]Cl3 = 4 ions
(b) [Co(H2O)5Cl]Cl2.H2O = 3ions
(c) [Co(H2O)4Cl2]Cl.2H2O = 2ions
(d) [Co(H2O)3 Cl3] 3H2O not possible
O.S = + 4 C.N. = 6
[Pt(NH3)4 Cl2]Cl2
O.S = +4 C.N = 6
H2[PtCl6]
Hexachloro platinic (IV) acid
O.S = +2 C.N. = 4
[Pd(NH3)4]Cl2
10
Answer
• Central metal atom releases e– according to its O.S.
• Central metal atom will provide vacant orbitals according to its coordination number.
• These vacant orbital undergo histrion and form coordinate s bond with donor atoms.
• Hybridisation state of central metal atom depends upon C.N. and nature of ligand M.M. dipole moment, isomerism etc. are also helpful in hybridisation prediction.
C.N = 2 sp linear
C.N = 3 sp2 Trigonal planar
C.N = 4 sp3 Tetrahedral
dsp2 square planar
C.N = 5 sp3d TBP
dsp3 (i) TBP
(ii) Square pyramidal (dx2y2)
C.N = 6 sp3d2 Octahedral (SBP)
d2sp3 Octahedral (SBP)
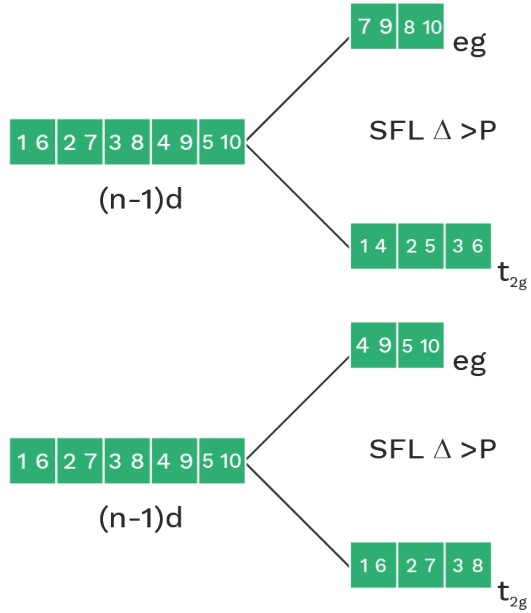
• In presence of strong field ligand (SFL), the pairing of (n–1)d e– is possible before hybridisation but this pairing is not possible in presence of WFL.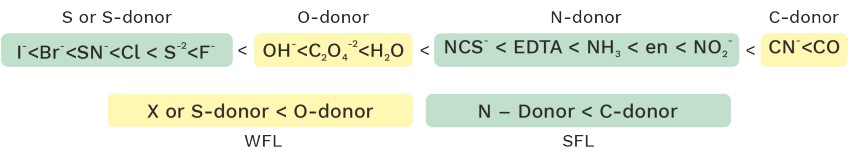
• All ligands acts as SFL with 4d and 5d series metal ions.
• P− acts as SFL with Ni+4
• H2O and C2O4−2 acts as SFL with CO+3
• NH3 act as WFL with Fe+2 and Mn+2 .
• H2O act as SFL with Cu+2 (C.N = 4)
(1) [Ni(CN)4]−2
O.S. = +2 C.N = 4 ligand = SFL
Ni = 3d84s2 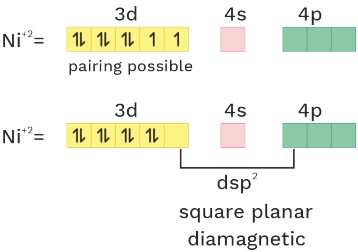
(2) [Ni(Cl4)]−2 or [Ni(SCN)4]−2
O.S = +2 C.N = 4 ligand = WFL
Ni = 3d84S2 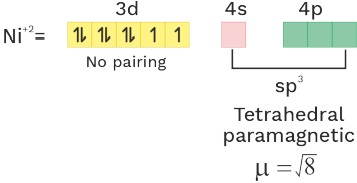
(3)[Fe(CO)5] D.M. = O
O.S. = 0 C.N. = 5 Ligand = SFL 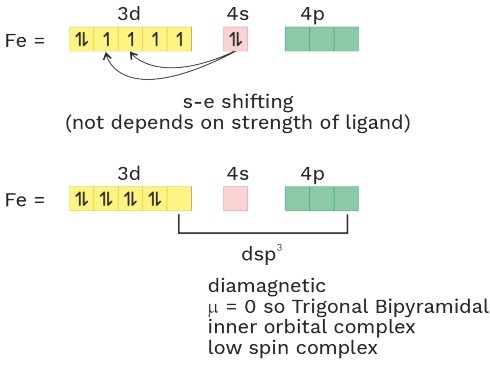
(4) [MnCl4]−2 sp3
O.S. = +2 C.N. = 4 Ligand = WFL 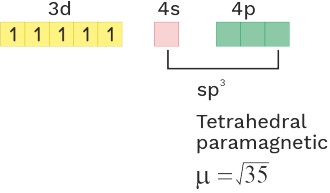
(5) [CuCl5]−3 sp3d
O.S. = +2 C.N = 5 Ligand = WFL 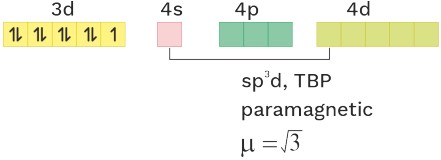
(6) [Ni(CO)4] or [Ni(CN)4]−4 sp3
O.S. = 0 C.N = 4 Ligand = SFL 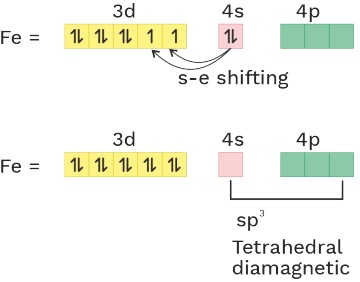
(7) [Fe(H2O)5(NO)]SO4
O.S = + 1 CN = 6 Ligand = WFL 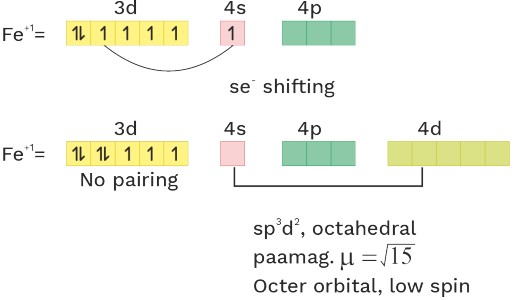
(8) [NiCl2(PPh3)2] Paramagnetic
O.S = + 2 CN = 4 Ligand = WFL = No pairing 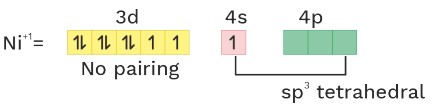
(9) [RhCl(PPh)3],[PdCl4]−2, [AgF4]− 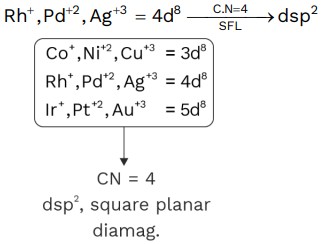
(10) [NiF6]−2 Ni+4 → F− act as SPL
O.S. = +4 CN = 6
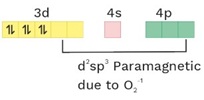
(11) [Fe(CN)5(O2−)]−4
O.S. = +2 CN = 6 Ligand = SFL 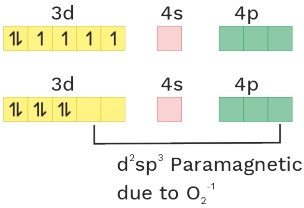
(12) [Fe(CO)4]−2
O.S. = −2 CN = 4 Ligand = SPL 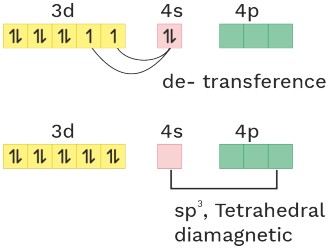
Drawbacks of VBT
(1) It can’t explain stability of complex compound
(2) It can’t explain colour of complex compound
(3) It can’t explain d e- transference
(4) It can’t explain pairing of (n-1)d e–
(5) It doesn’t give any criteria for classification of SFL and WFL.
CFT for octahedral complex
dxy , dyz , dzx = t2g
dz2 , dx2 – y2 = eg 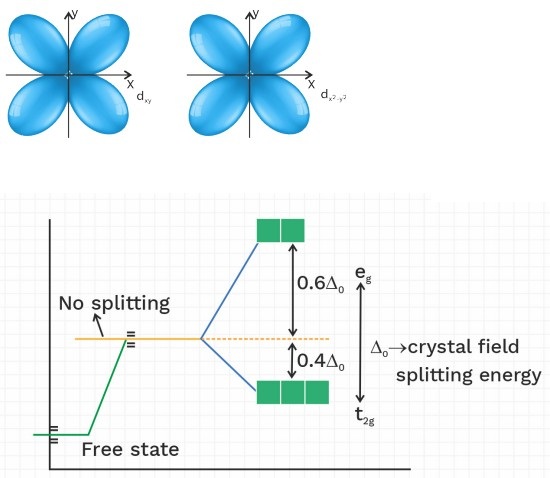
11
Answer
In crystal field theory bonding between metal and ligands is purely electrostatic.
Ligands are considered as negative point charges.
What Happens When Ligands Approach A Metal 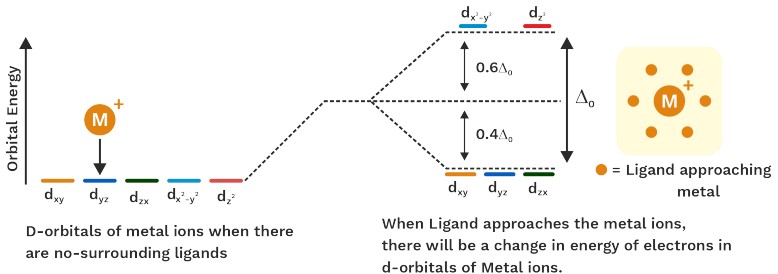
Orbital Splitting In Octahedral Complexes 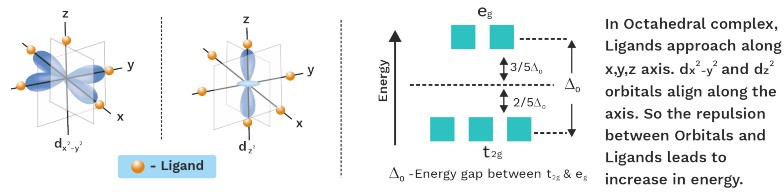
Orbital Splitting In Tetrahedral Complexes 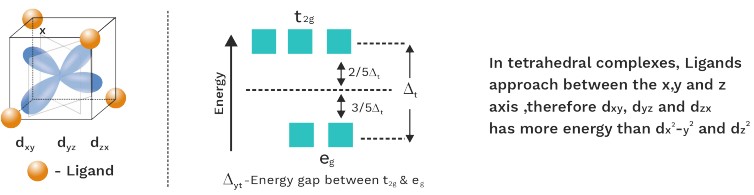
Strenght Of Ligands
CO ≈ CN− > PPH3 > NO2− > NH3 > pyridine > CH3CH > NCG− > H2O ≈ C2O42− > OH− > NCO− > F− > Cl > SCN > S2− > Br− > l− > O22− .
12
Answer
Electronic configuration of (n – 1) de− of CMA (after splitting) depends upon 2 type of energies are :
(1) ∆0(2) Pairing energy (P)
There are two conditions
(1) In presence of SFL ∆0 > P
(2) In presence of WFL ∆0 < P 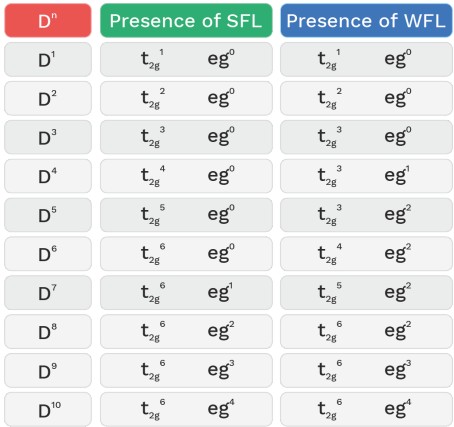
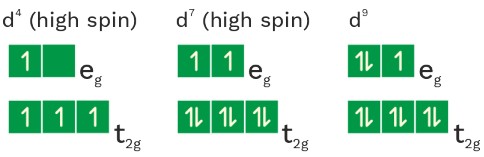
Hybridisation State and Magnetic Nature 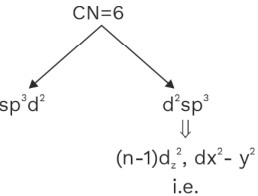
Configuration of metal in 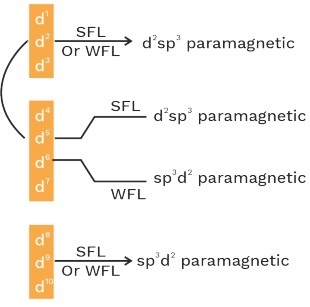
Factor affecting splitting energy
(1) ∆0 ∝ charges of central metal atom
(2) ∆0 ∝ Zeff of central metal atom ![]()
(3) ∆0 ∝ strength of ligand
(4) Geometry of complex

Examples, ![]()
H2O acts as strong ligand with Co+3, so splitting energy for (II) is higher than (I)
(ii) [Fe(H2O)6]+3 < [Ru(H2O)6]+3 < [Os(H2O)6]+3 ∆0
Fe is a 3d series elements whereas Ru and Os are 4d and 5d transition elements, so H2O is weaker ligand but it acts as strong ligand with higher d series transition element.
(iii) [CrCl6]−3 <[Cr(NH3)6]+3 <[Cr(CN)6]−3 ∆0
Ligand strength for CN− > NH3 >Cl− ![]()
(I) is tetrahedral and (II) is octahedral complex
∆t > ∆0
Stability of complex compound 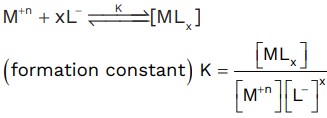
K↑, stability↑
Stability ∝ charge of CMA
∝ Zeff of CMA
∝ Strength of ligand
∝ chelation effect 
Stability order :
(1) [Fe(CN)6]−4 <[Fe(CN)6]−3
O.S = +2 O.S. = +3
(2) [Co(H2O)6]+3 <[Rh(H2O)6]+3 <[Ir(H2O)6]+3 Zeff ↑
(3) [NiCl6]−4 <[Ni(NH3)6]+2 <[Ni(CN)6]−4 ligand strength
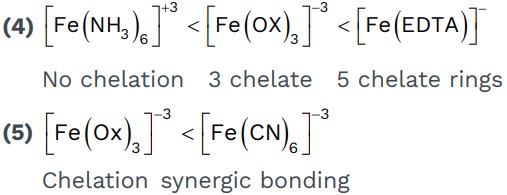
(6) Irving williams series
Mn+2
13
Answer
Extra stabilisation (released energy) due to splitting in comparison to no splitting
[Fe(CN)6]−4 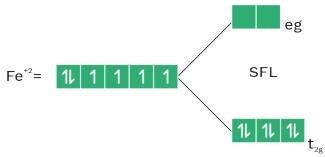
Priority to 2P = N, New pairs
CFS = −0.4∆0 × 6 + 0.6∆0 × 0
= −2.4∆0
Total pairs
CFSE = 0.4∆0 nteg + 0.6 ∆0 nteg + xp = new pairs
CFT for Tetrahedral complex 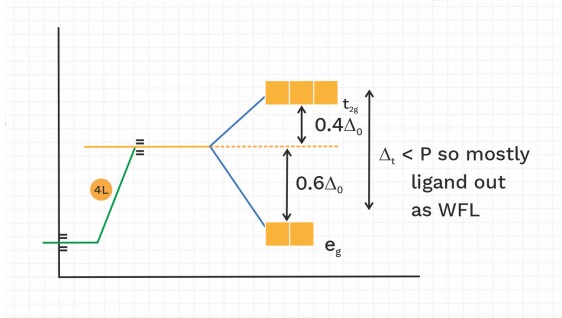
14
Answer
(a) d – d Transition
• Colour of complex compound is due to d – d transition
• Ag. Solution of Ti+3 is violet. 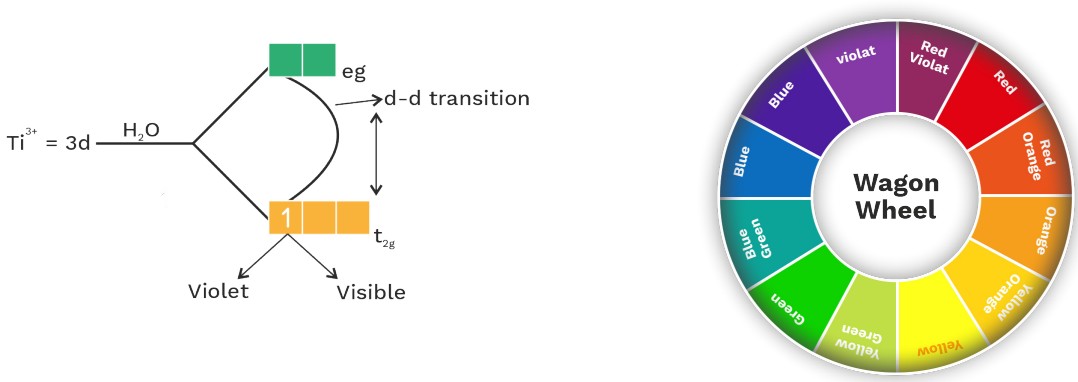
• Colour of complex compound depends upon spilitting energy.
• Complex become colourless in absence of ligand filled (no ligand, no splitting, no transition)
• Colour of f – block compounds is due to f – f transition.
Examples,
(i) [Ti(H2O)6]+ become colourless on heating due to removal of water molecules.
(ii) Anhydrous CuSO4 is colourless but hydrated CuSO4 is blue (test of moisture)![]()
(b) Charger transfer
Colour of some compound is due to charge transfer (e− transition) from anion to metal ion. 
(c) Polarisation
• Colour of same compound can be explained on the basis of polarisation.
• Polarisation increase, possibility of finding colour increase
Examples,
(1) AgF colourless but Agl yellow.
(2) PbF2 colourless but Pbl2 yellow.
(3) HgF2 colourless but Hgl2 Red.
(4) ZnS colourless but Cds yellow. HgS Black.
Colour of Co – Ordination compounds 
Why we are seeing Blue, Why Not Red or Green?
Let’s see what happens inside the solution containing co – ordination compound when we cast a white light, on it 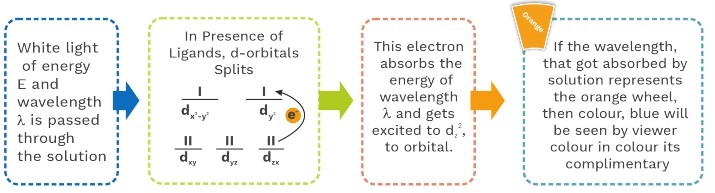
Colours shown by metals in various oxidation states 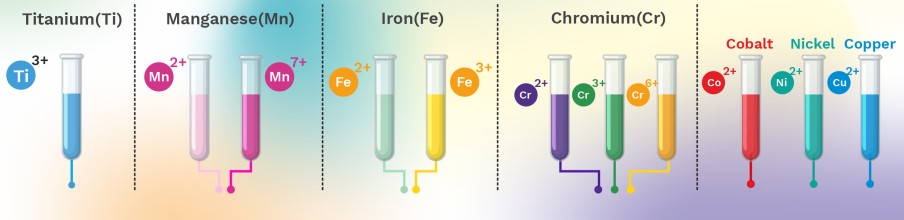
15
Answer
• Same molecular formula but different structural formula
(1) Coordination Isomerism
It arises due to exchange of ligands between complex cation and complex anion.
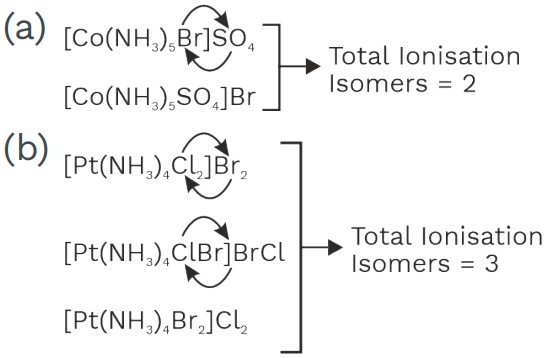
(2) Ionisation Isomerism
• Structural isomer which give differ ions in aqueous solution
• Ionization isomerism is the result of the exchange of groups or ions between the coordinating sphere and the ionization sphere.
Examples,
(3) Hydrate Isomerism
• It is a special type of ionisation isomerism in which number of water molecules differ in coordination sphere/outside region.
Examples, 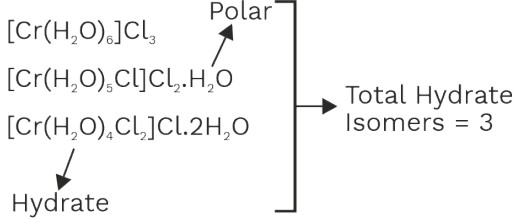
(4) Linkage Isomerism
• It arise due to presence of ambidentate ligand.
Examples,
[Co(NO2)(NH3)5]Cl2
Pentaamminenitrocobalt (III) chloride
(5) Polymerisation Isomerism
• Complexes which have differ molecular formula but have same empirical formula. It is not a true isomerism.
Example,
[PtCl2(NH3)2] and [Pt(NH3)4][PtCl4]
16
Answer
• Tetrahedral complexes don’t show ‘Geometrical Isomerism’.• Square planar complexes can show ‘Geometrical Isomerism’.
• Any two identical ligands at 180° show Trans.
• All identical ligands at 90° show Cis.
Examples,
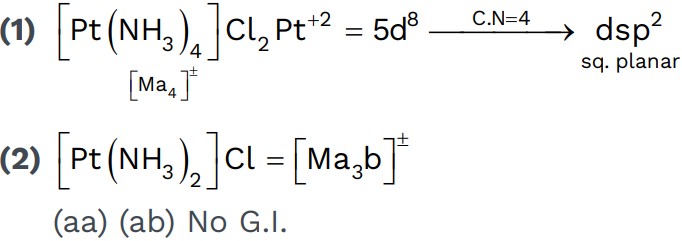
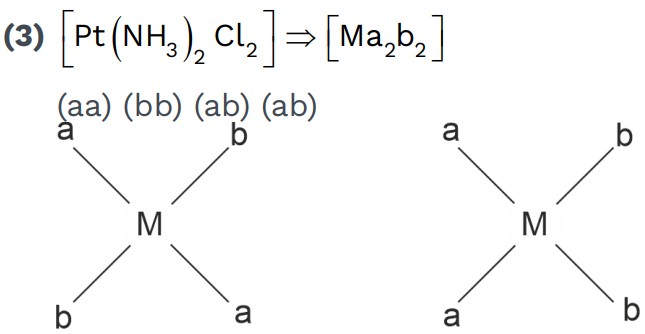
Is[Pt(NH3)2 Cl2 B] – it is used as anticancer agent.
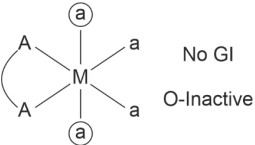
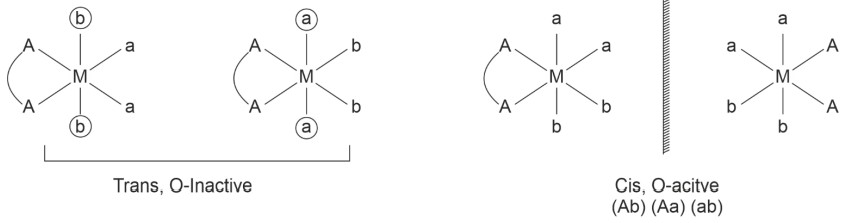
• Geometrical and Optical Isomerism in octahedral complexes

(4) [Pt(en)Cl4] ⇒ [M(AA)a4]±
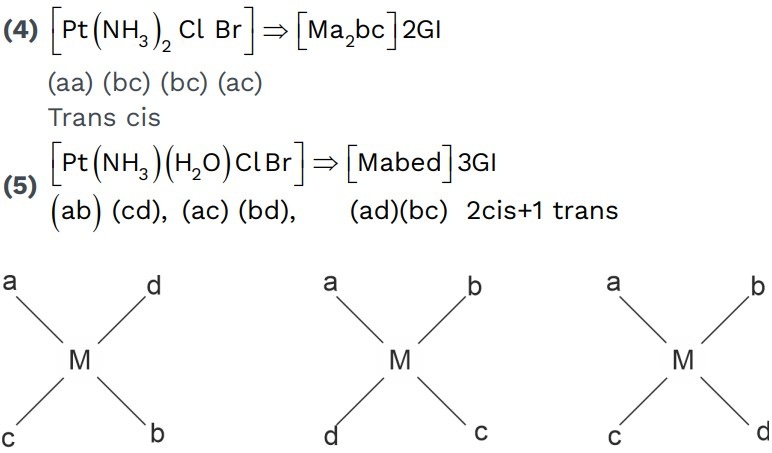
(5) [Pt(en)Cl2Br2] = [M(AA)a2b2]
GI = 3
O – active = 3
S.I = 4
(6) [M(AA)2ab]±
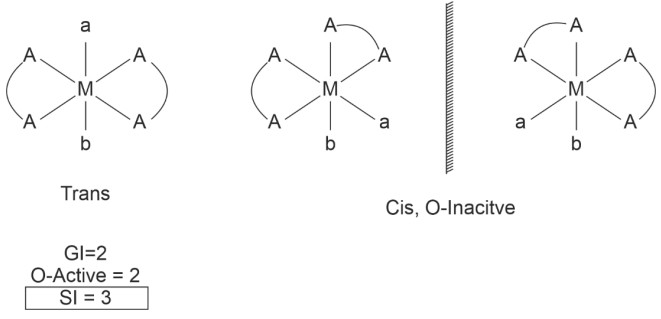
17
Answer
(1) Test of Ni+2 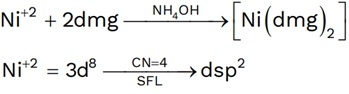
Square planer diamagnetic 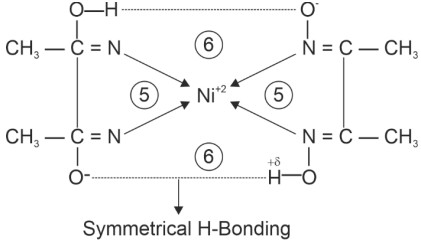
(2) Test of Fe+2, Fe+3 and Cu+2 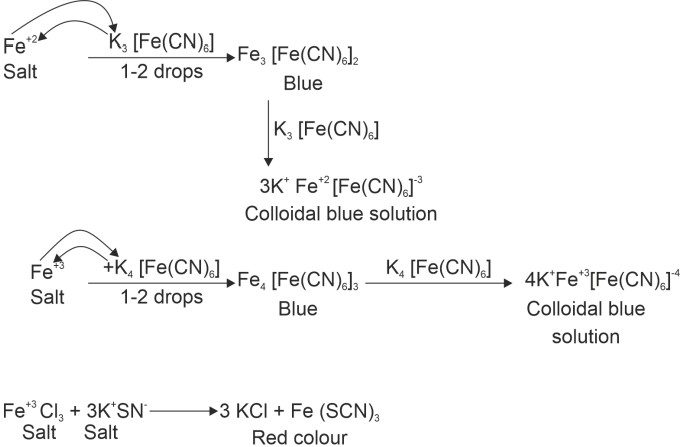
(3) Test of sulphide ion ![]()
S−2 + N+O → NOS
(4) Separation of hydroxides or oxides 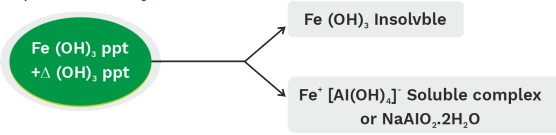
(5) If excess amount of KCN is added into CuSO4 solution then insoluble CuCN is formed which later into soluble complex.
(6) AgCl or AgBr ppt (not Agl) are soluble in NH3 or NH4OH ![]()
(7) [EDTA]−4 is used for estimation and removal of hardness of H2O.
Ca+2 + EDTA−4 → [Ca(EDTA)]−2
(8) Wilkinson catalyst [RhCl(PPh)3] is used for hy – drogenation of alkene.
(9) Biological importance : -
- Chlorophyll → Mg
- Vit – B12 → Co
- Carboxypeptide → Zn
- Plastocynin → Cu
- Insolin → Zn
- Haemoglobin → Fe+2
- Hyoglobin → Fe+2
18
Answer
Compounds in which carbon atom of hydrocarbon part is directly bonded with metal or metalloid.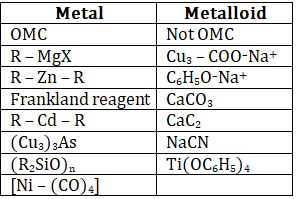
(1) σ Bonded OMC
(C2H5)4Pb T.E.L used as anti knocking agent [TiCl4 + (C2H5)3 Al] Ziggler natta catalyst(Heterogenous catalyst) = Used for polymerisation of alkene.
(2) π Bonded OMC
Presence of π donor ligand
[Cr(η6 – C6H6)2], [Fe(η5 – C5H5)2]
(3) σ and π bonded OMC
Presence of synergic bonding
[Ni(CO)4] ∙[Fe(CO)5]
Trans effect
K[PtCl3(η2 – C2H4)]
square planar diamagnetic
John Taller effect
Distortion in octahedral geometry due to unsymmetrical e− cloud in eg set of orbitals.