NCERT Revision Notes for Chapter 3 Chemical Kinetics Class 12 Chemistry
CBSE NCERT Revision Notes1
Answer
- Chemical kinetics : It is the branch of chemistry which deals with the study of reaction rates and their mechanisms.
- Rate of a reaction: The rate of a reaction can be defined as the change in concentration of a reactant or a product in unit time. For the reaction,
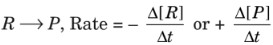
Units of rate: Concentration time–1e., mol L–1 s–1 or atm s–1 for gaseous reactions. - Average rate of reaction: It is the average value during a large time interval.
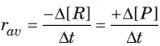
- Instantaneous rate of reaction: It is the rate of a reaction at a particular instant of time i.e., when ∆t approaches zero.
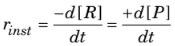
2
Answer
- Concentration: Greater the concentrations of the reactants, faster is the rate of reaction.
- Physical state of reactants : Reactions involving gaseous reactants are faster than reactions containing solid and liquid reactants.
- Temperature : The rate of reaction increases with increase of temperature. For most of the reactions, rate of reaction becomes almost double with 10° rise of temperature.
- Presence of catalyst : A catalyst generally increase the speed of a reaction.
- Surface area of reactants: For a reaction involving a solid reactant or catalyst, the greater is the surface area, the faster is the reaction.
- Presence of light : Photochemical reactions take place in the presence of light only.
- Activation energy: Lower the activation energy faster is the reaction.
3
Answer
The equation that correlates the rate of reaction with concentration of reactants is known as rate law.
- For a simple reaction, A + B → C + D
Rate = k[A][B] where k is the rate constant which is equal to the rate of reaction when concentration of each of the reactant is unity. - For a simple reaction, aA + bB → cC + dD
Rate = k[A]x[B]y and order of reaction = x + y
4
Answer
- Half – life or reaction: The time in which the concentration of a reactant is reduced to one half of its initial concentration is called half – life of the reaction.
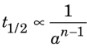 where n is the order of the reaction.
where n is the order of the reaction.
5
Answer

6
Answer
- Zero order reaction
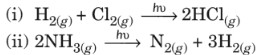
- First order reactions
(i) NH4NO2 → N2 + 3H2O
(ii) 2N2O5 → 4NO2 + O2 - Second order reactions
(i) 2HI → H2 + I2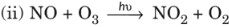
- Third order reactions
(i) 2NO + O2 → 2NO2
(ii) 2NO + Br2 → 2NOBr - Fractional order reactions
(i) H2 + Br2 → 2HBr
Rate = k[H2][Br2]½ , Order = 1.5
(ii) CH3CHO → CH4 + CO
Rate = k[CH3CHO]3/2 , Order = 1.5
7
Answer
Plots of rate vs concentration: 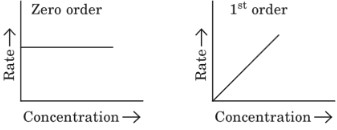
Plots of integrated rate equations: 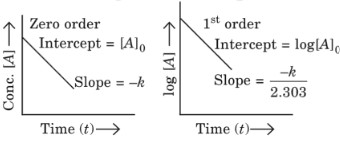
8
Answer
Those reactions which are not truly of the first order but under certain conditions become reactions of the first order are called pseudo first order reactions. e.g.,
Acid hydrolysis of ethyl acetate: 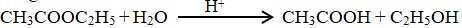
Rate = k’[CH3COOC2H5]
= k[CH3COOC2H5] where, k = k’(H2O]
Acid catalyzed inversion of cane sugar:
![]()
Rate = k[C12H22O11]
9
Answer
Arrhenius equation:
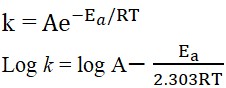
where, k = Rate constant,
A = Pre – exponential factor
(frequency factor),
Ea = Activation energy,
T = Temperature 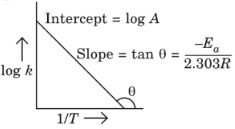
If k1 is rate constant at T1 and k2 at T2, then we get 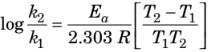
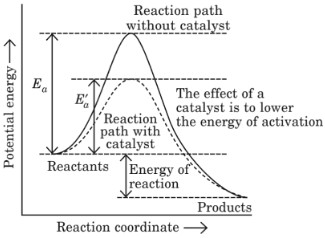
Activation energy: The minimum amount of energy required by reactant molecules to participate in a reaction is called activation energy (Ea). 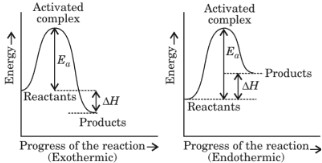
Activation energy = Threshold energy – Average kinetic energy of reacting molecules
Activation energy (Ea) = E(activated complex) – E(ground state)
∆H = Activation energy of forward reaction – Activation energy of backward reaction.
10
Answer
This theory is based on assumption that collisions between reactant molecules are responsible for the formation of product molecules, and rate of reaction is governed by the collision frequency (Z), (Z = no. of collisions per second per unit volume of reaction mixture).
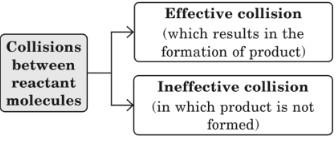
Two factors decide whether a collision will be effective or not and these are:
- Energy factor: For an effective collision, the colliding molecules must possess a certain minimum (or more) value of energy and this minimum value of energy is called threshold energy.
- Orientation factor: Only those collisions among active molecules are effective which are properly oriented.
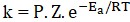
Where, p = orientation factor or probability factor.
Z = collision frequency between molecules.