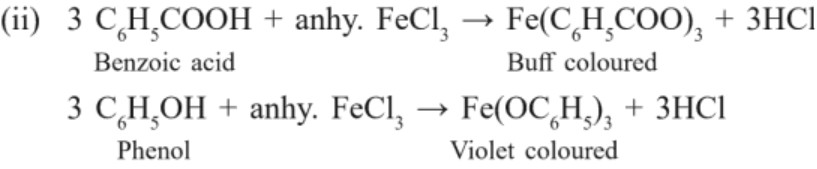NCERT Revision Notes for Chapter 8 Aldehydes, Ketones and Carboxylic Acids Class 12 Chemistry
CBSE NCERT Revision Notes1
Answer
C –O bond is polarized due to higher electronegativity of oxygen relative to carbon. Hence, carbonyl carbon is an electrophilic (Lewis acid) and carbonyl oxygen is a nucleophilic centre (Lewis base).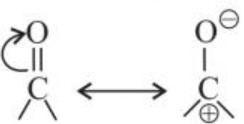
2
Answer
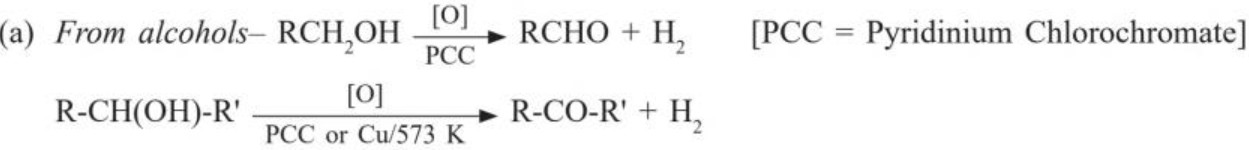
(b) From hydrocarbons
(i) By ozonolysis of alkenes :
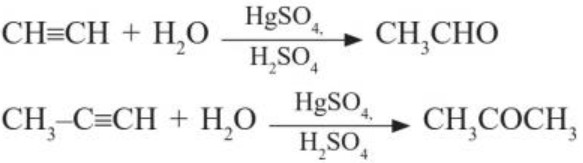
(ii) By hydration of alkynes :
3
Answer
(a) Rosenmund reaction :[BaSO4 = poisons the catalyst to prevent further reduction of aldehyde]
(b) (i) Stephen’s reduction :
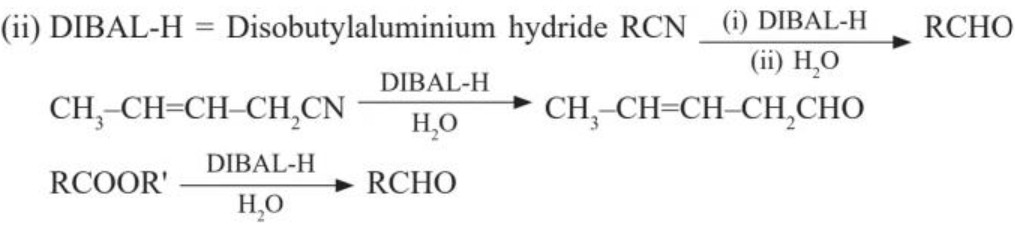
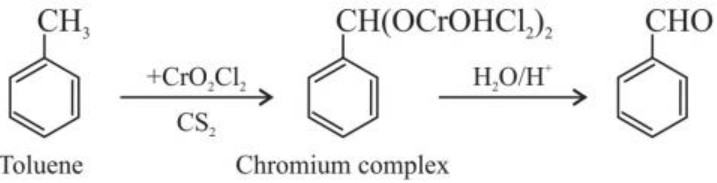
(c) From hydrocarbons :
(i) Etard reaction : CrO2Cl2 = Chromyl Chloride (red vapours)
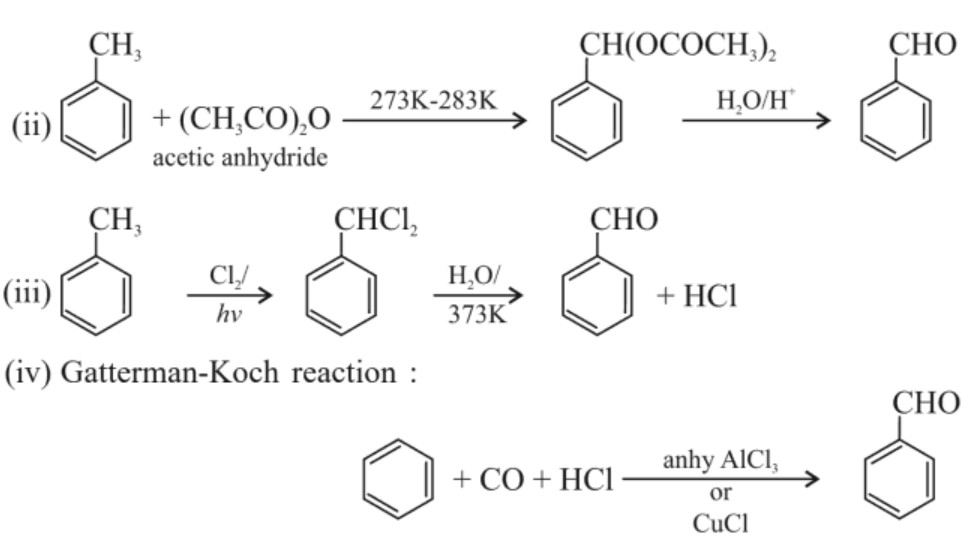
4
Answer
(a) From acyl chlorides : Grignard reagent cannot be used directly for the preparation of ketone as it will form alcohol with acyl chloride and required product will not be obtained. Hence, Grignard reagent is treated with CdCl2 (cadmium chloride) to form dialkyl cadmium which reacts with acyl chloride to form ketone.2RMgX + CdCl2 → R2Cd + 2Mg(X)Cl
(b) Friedal – Craft acylation reaction :
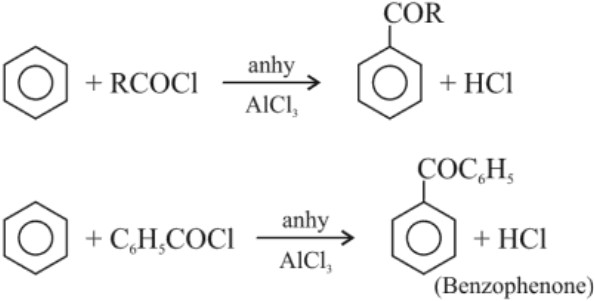
(c) From nitriles :
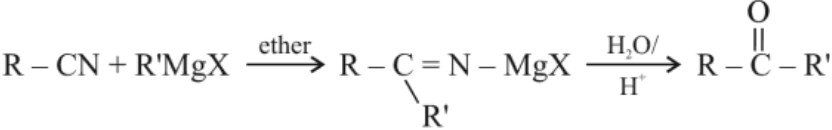
5
Answer
(a) Boiling point and melting point : Alcohols > Aldehydes > Ethers > Hydrocarbons− Alcohols are associated with intermolecular Hydrogen bond, Aldehydes and ketones have dipole – dipole interactions but they do not have intermolecular H – bonding. Ethers and hydrocarbons are associated with weaker van der Waals’ forces.
(b) Solubility : Aldehydes and ketones form hydrogen bonds with water and are soluble in water. Their solubility decreases with increase in number of carbon atoms in alkyl chain.
6
Answer
(I) Nucleophilic addition reactions :Mechanism – Nucleophilic attacks the electrophilic carbon atom of carbonyl group. Hybridisation changes from sp2 to sp3. The intermediate formed captures proton from reaction medium.
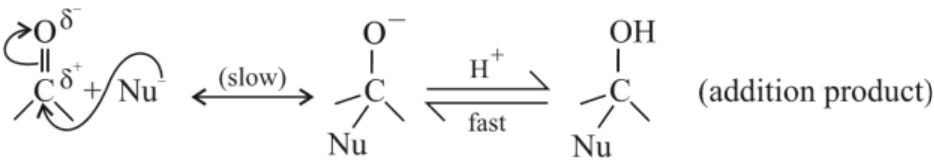
Note : Aldehydes are more reactive than ketones towards nucleophilic addition reaction because –
(a) Ketones contain two alkyl groups which cause steric hinderance to the approach of nucleophile towards carbonyl carbon while aldehydes contain only one alkyl group causing comparatively less steric hinderance.
(b) The two alkyl groups present on ketone reduce the electrophilicity of carbonyl due to their +I effect.

Note : Benzaldehyde is less reactive towards nucleophilic addition than aldehyde because the carbon atom of carbonyl group of benzaldehyde is less electrophilic than in case of aldehyde. This occurs due to resonance in benzaldehyde.
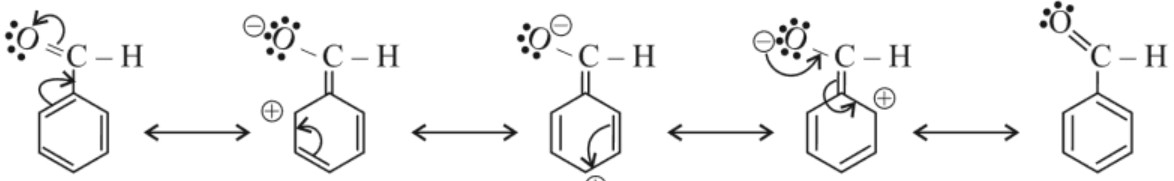
Reactions :
(a) Addition of HCN :
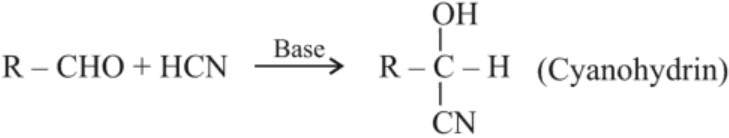
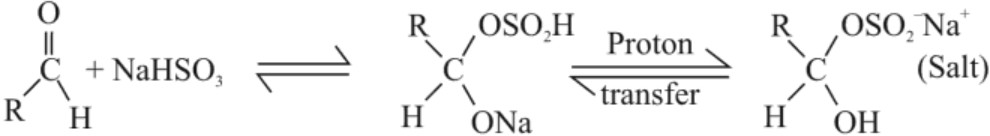
(b) Addition of Sodium hydrogensulphite :
(c) Addition of Grignard reagent :
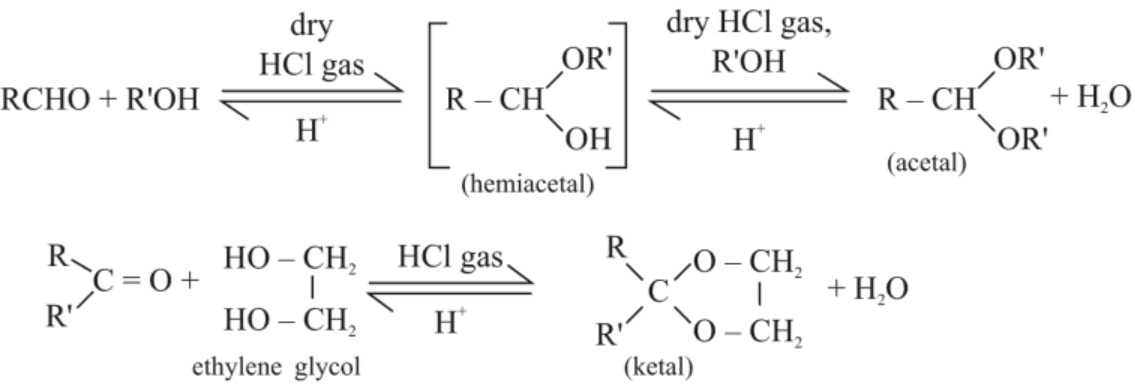
(d) Addition of alcohols :
(e) Addition of ammonia and its derivatives :
RCHO + NH3 →R – CH(OH) – NH2 → R – CH = NH (imine) + H2O
Note : Ammonia derivatives requires slightly acidic medium for the reaction with aldehydes and ketones pH = 4 to 5) because ammonia derivatives are weak nucleophiles and we cannot increase the reactivity of nucleophile but we can increase the acceptability of carbon of carbonyl group by providing acidic medium.
(i) Reaction with amine :
(ii) Reaction of hydrazine :
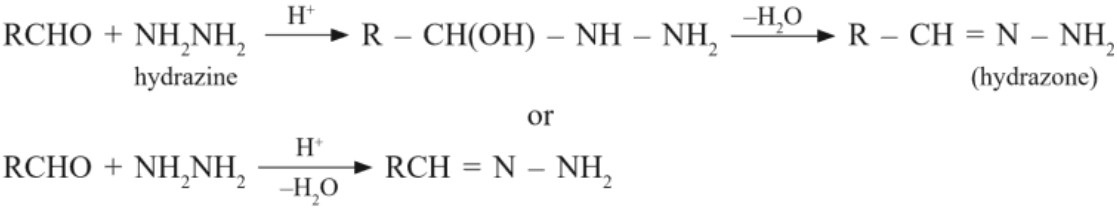
(iii) Reaction with semicarbazide :
Only one –NH2 group can act as nucleophile in semicarbazide because other –NH2 group is involved in resonance with carbonyl group. So, its lone pair is not available for donation (or reaction).
(iv) Reaction with Hydroxylamine:
(v) Reaction with phenylhydrazine :
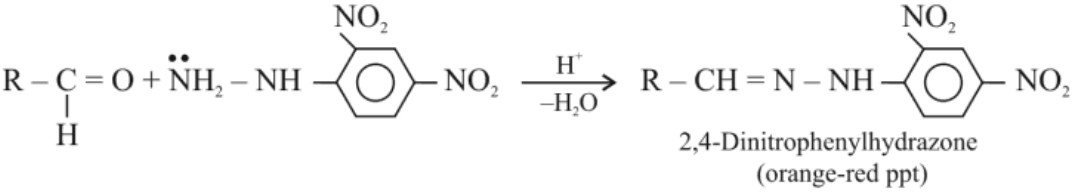
(vi) Reaction with 2, 4 – Dinitrophenylhydrazine (2, 4 – DNP): Test for aldehydes and ketones both
(II) Reduction :
(a) Reduction to alcohols : Reducing agents = LiAlH4 or NaBH4 or H2/Pd
(b) Reduction to hydrocarbons
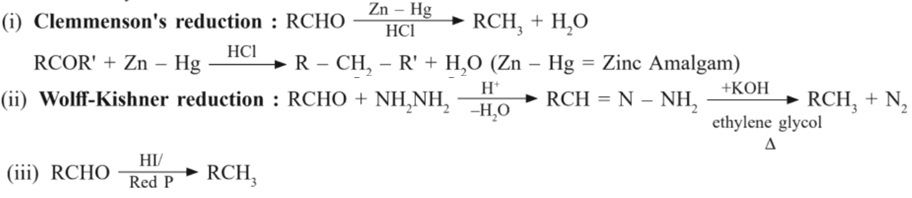
(III) Oxidation :
Oxidizing agent = Concentrated HNO3 or alkaline KMnO4 or acidified K2Cr2O7 or mild oxidising agents like Tollens’ reagent, Fehling’s reagent or Benedict’s reagent.
(Ketone requires drastic conditions for oxidation)
(b) Tollens’ test (for aldehydes only): Tollens’ reagent = Silver ammoniacal nitrate
(c) Fehling’s test : (for aldehydes only – only for aliphatic aldehydes. Aromatic aldehydes do not respond to this test)
Fehling’s reagent = Fehling solution A (aqueous CuSO4) + Fehling solution B (Sodium potassium tartarate or Rochelle’s salt)
(d) Haloform test or lodoform test : Given by compounds containing one methyl group linked to carbonyl carbon atom (methyl ketones : −CO – CH3) or by those compounds which on oxidation form methyl ketones.
Iodoform test : NaOH + I2 → NaOI + HI
R – CO – CH3 + NaOI → CHI3 + 3HI + RCOONa or
Compounds which will give positive iodoform test are : CH3COCH3 (acetone or propanone), CH3CHO (ethanol), CH3CH2OH (ethanol), CH3COCH2CH2CH3 (pentan – 2 – one), C6H5–COCH3 (Acetophenone), etc.
(iv) Reactions due to α–hydrogen :
α–hydrogen of aldehydes and ketones are acidic in nature due to the strong electron withdrawing effect of carbonyl group and resonating stabilisation of conjugate base.
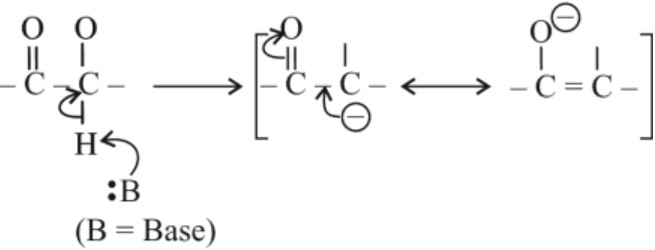
(a) Aldol Condensation : Aldehydes and ketones having atleast one α–hydrogen undergo a reaction in presence of dilute alkali as catalyst to form β–hydroxyl aldehydes (aldol) or β–hydroxyl ketones (ketols). This reaction is known as aldol condensation. On heating aldol or ketol, α, β–unsaturated carbonyl compounds are formed.
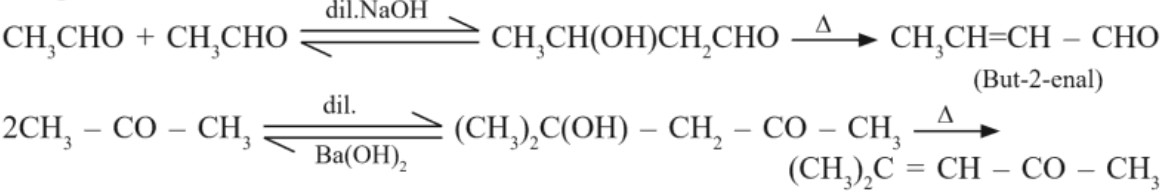
(b) Cross – aldol condensation : When aldol condensation is carried out with two different aldehydes or ketones, it is called as Cross – aldol condensation. If both of the reactants contain α–hydrogen, then we get mixture of four products.
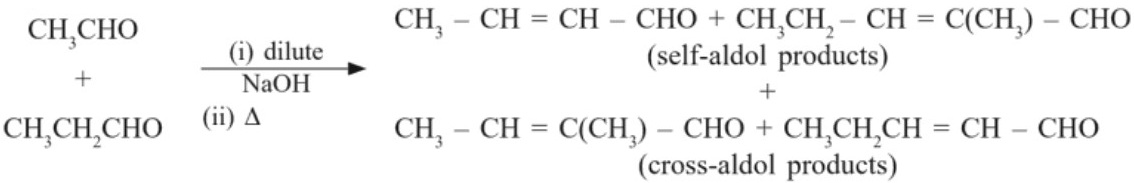
Ketones can also be used as one component in the reaction :
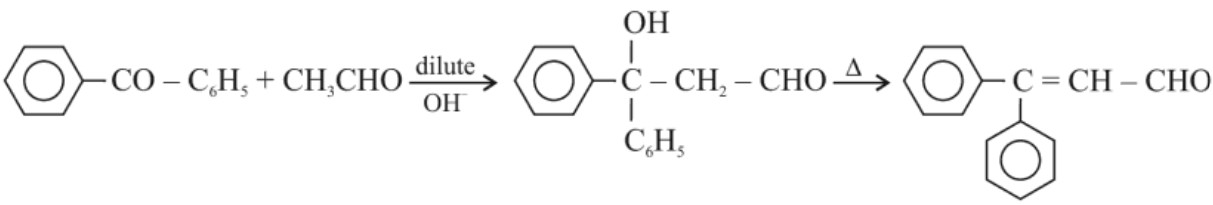
(v) Other reactions :
(a) Cannizzaro reaction : Aldehydes which do not have α–hydrogen undergo self – oxidation and reduction (disproportionation) reaction on treatment with concentrated alkali. In this reaction, one molecule of aldehyde is reduced to alcohol and another is oxidised to carboxylic acid salt.
HCHO + HCHO + Conc. KOH → CH3OH + HCOOK
2C6H5CHO + conc. NaOH → C6H5COONa + C6H5CH2OH
(b) Electrophilic substitution reactions : Aldehydes and ketones are meta – directing and deactivating groups attached to benzene. Election withdrawing group (–CO–) would cause electron density at meta position due to resonance and electrophile with attack at meta – position.
7
Answer
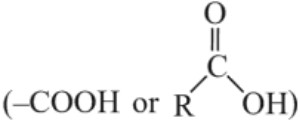
–COOH group is involved in resonance
8
Answer
(a) Oxidation of alcohols :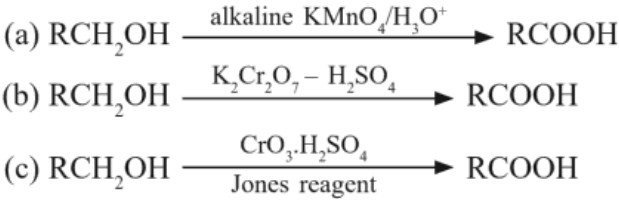
(b) From alkyl benzene
(c) From alkenes
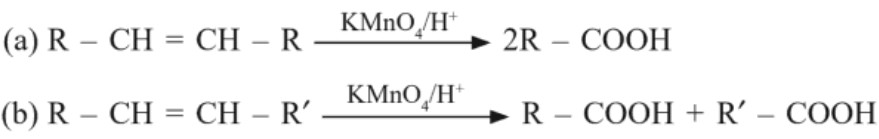
(d) From nitriles
(e) From Grignard reagent :
(f) From acid derivatives
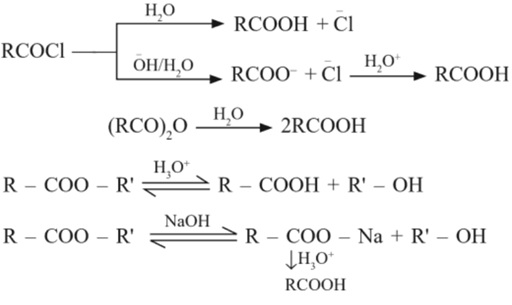
9
Answer
(a) Aliphatic carboxylic acids having even number of carbon atoms have higher boiling point and melting point than odd number of carbo atoms because even no. of C atoms containing carboxylic acids have terminal methyl group and carboxylic acids in zig – zag structure which are far apart from each other and have less repulsion and more stability due to which they fit snugly into their lattice which in case of odd no. carbons containing carboxylic acids, terminal methyl group and carboxylic acid group are closer to each other and have more repulsion, less stability.(b) Carboxylic acid form dimer because of H – bonding (intermolecular) or association in vapour or in aprotic solvents

(c) They are soluble in water utpo 4 carbon atoms due to intermolecular H – bonding with water. Solubility decreases with increase in number of carbon atoms.
10
Answer
![]()
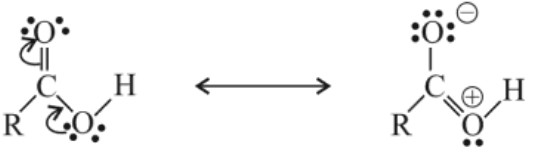
(i) Carboxylic acids are acidic because carboxylate ion formed is stable and hence carboxylic acid has greater tendency to ionize to form stable carboxylate ion due to its equivalent resonating structures in which negative charge is dispersed in the structure. 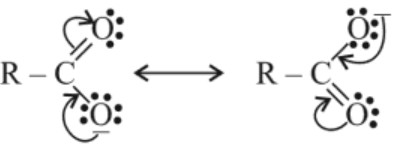
2RCOOH + 2Na → 2RCOONa + H2
RCOOH + NaHCO3 → RCOONa + H2O + CO2
Strength of acid → (pKa = −log Ka)
∗Ka (Dissociation Constant) increases, pKa decreases, stronger is the acid.
(ii) Effects of substituents on acidic strength of acids:
• e– releasing groups like alkyl groups destabilize the carboxylate ion due to their +I effect and weakens the acid, i.e., carboxylic acid will not be able to release H+ ions easily when it is attached to e– releasing group.
• e– withdrawing groups like halogens, −NO2 stabilize the carboxylate anion by dispersing the negative charge on carboxylate ion and strengthens the acid. ![]()
Note : Order of acidity : CCl3COOH > CHCl2COOH > CH2ClCOOH > HCOOH > Cl – CH2 – CH2 – COOH > C6H5COOH > C6H5CH2COOH > CH3COOH > CH3CH2COOH > (CH3)2CH – COOH
• Direct attachment of groups such as phenyl or vinyl to the carboxylic acid increases the acidic character. This is because of greater electronegativity of sp2 hybridized carbon to which carbonyl group is attached.
(b) Chemical Tests :
(i) RCOOH + NaHCO3 → RCOONa + H2O + CO2(gas) [brisk effervescence]
ROH + NaHCO3 → no reaction