NCERT Revision Notes for Chapter 9 Amines Class 12 Chemistry
CBSE NCERT Revision Notes1
Introduction
Answer
Alkyl or aryl derivatives of ammonia are regarded as amines. These are obtained by replacing one or more hydrogen atoms by alkyl or aryl groups. Amines are classified as primary, secondary or tertiary depending upon whether one, two or three atoms of hydrogen have been replaced by alkyl or aryl groups.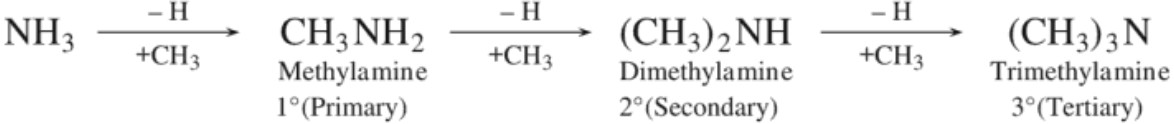
Thus, characteristic functional groups for 1°, 2° or 3° amines are :
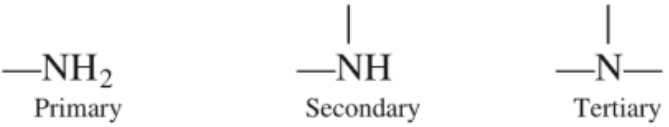
Amines are said to be ‘simple’ when all the alkyl or aryl groups are the same, and ‘mixed’ when they are different. There is another class of compounds wherein all the four hydrogen atoms of an ammonium salt have been replaced by alkyl or aryl groups. Such compounds are named as quaternary ammonium salts.
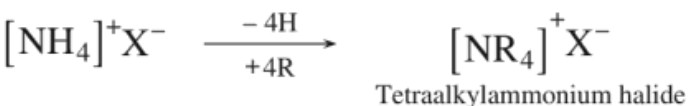
2
Preparation of Amines
Answer
Amines are prepared by the following methods :(a) Reduction of nitro compounds :
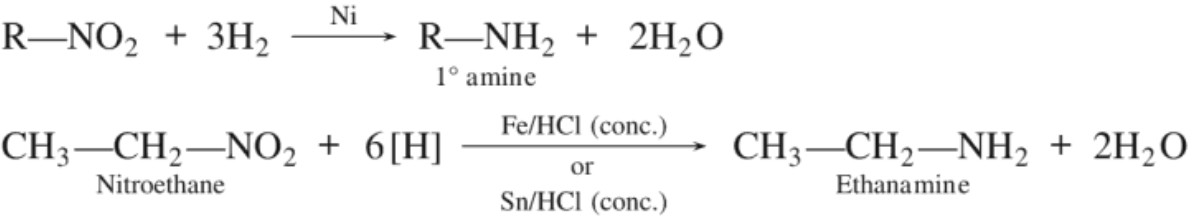
(b) Ammonolysis of alkyl halides :
If alkyl halide is in excess, the amine formed further reacts with alkyl halide to form 2°, 3° amines and finally quaternary ammonium salts.
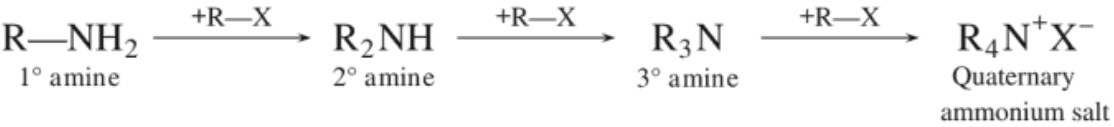
(c) Reduction of nitriles :
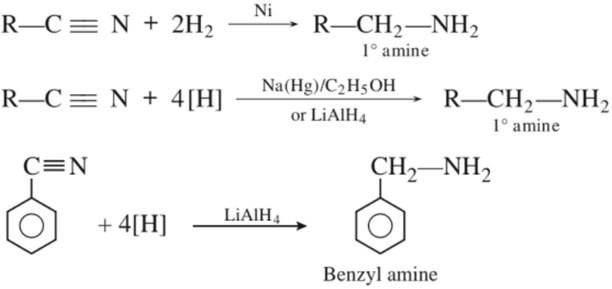
(d) Reduction of amides :
(e) Hoffmann’s bromamide degradation reaction :
(f) Gabriel phthalimide synthesis :
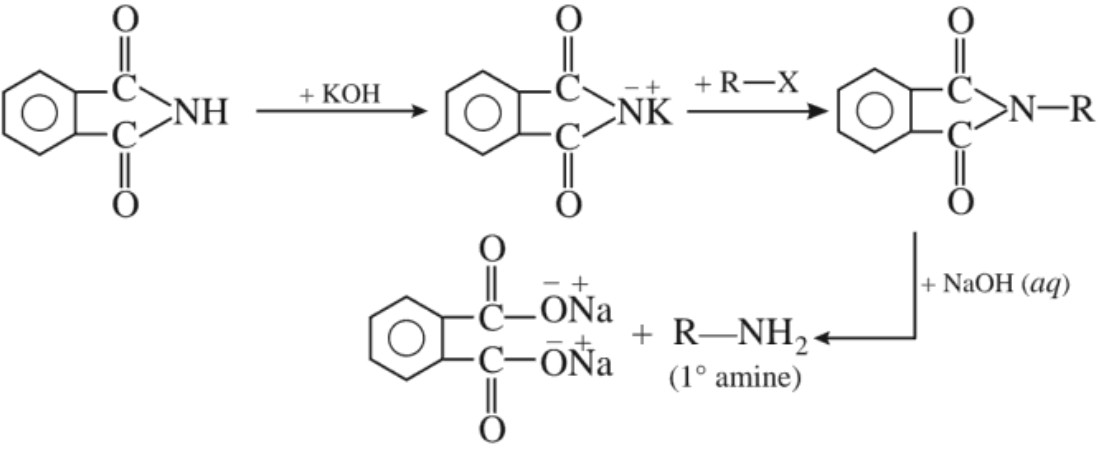
Aromatic amines cannot be prepared by this method because aryl halides do not undergo nucleophilic substitution with the anion formed by phthalimide.
Preparation of Aniline :
(a) From nitrobenzene
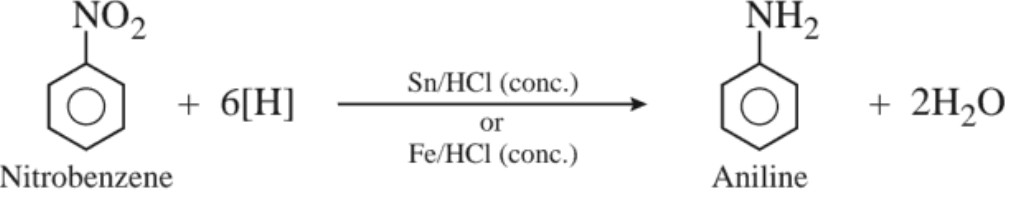
(b) By Hoffmann bromamide degradation reaction
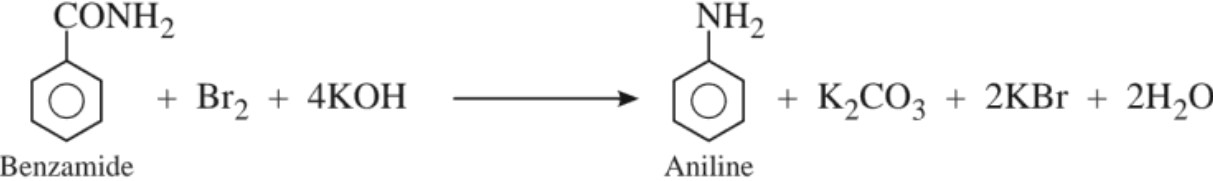
3
Chemical Properties of Amines
Answer
(a) Alkylation :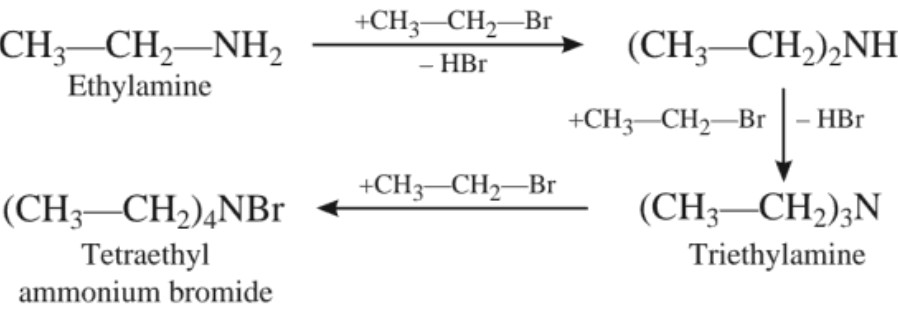
(b) Acylation :
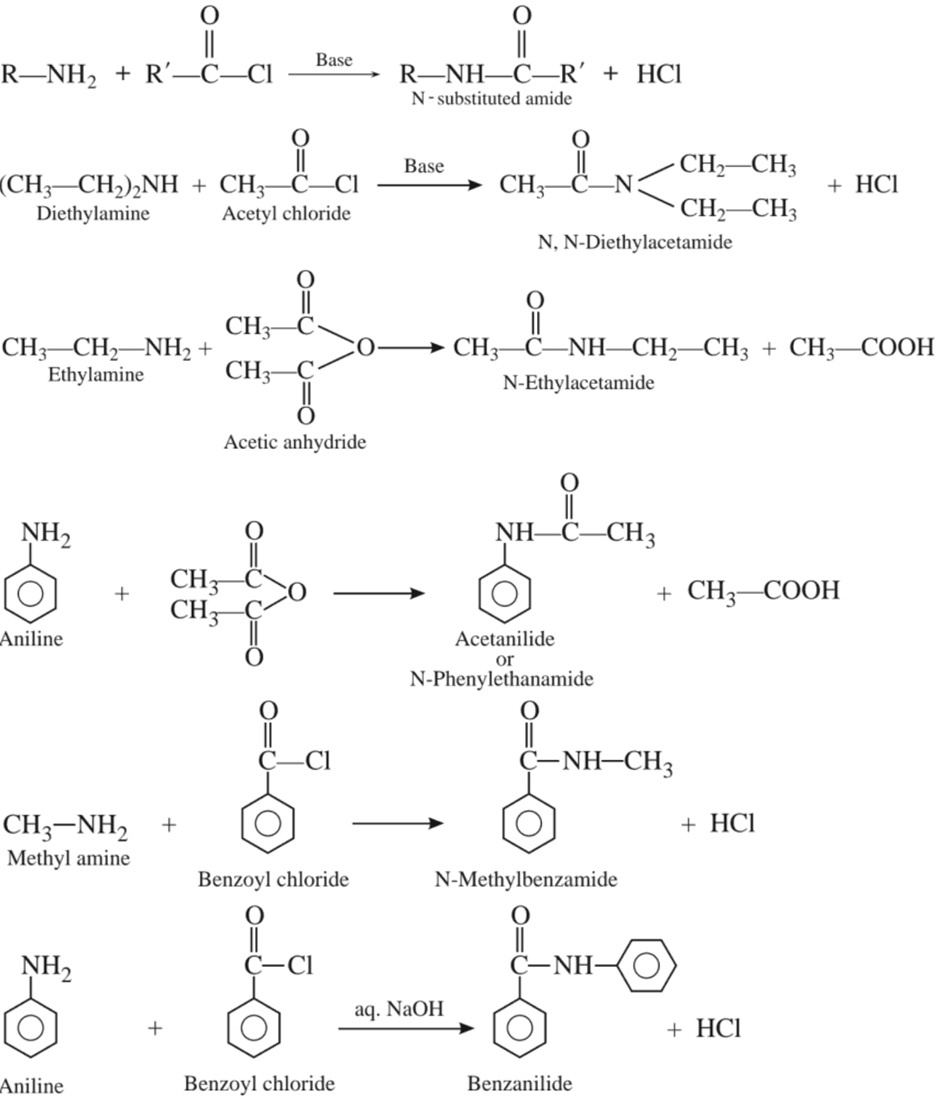
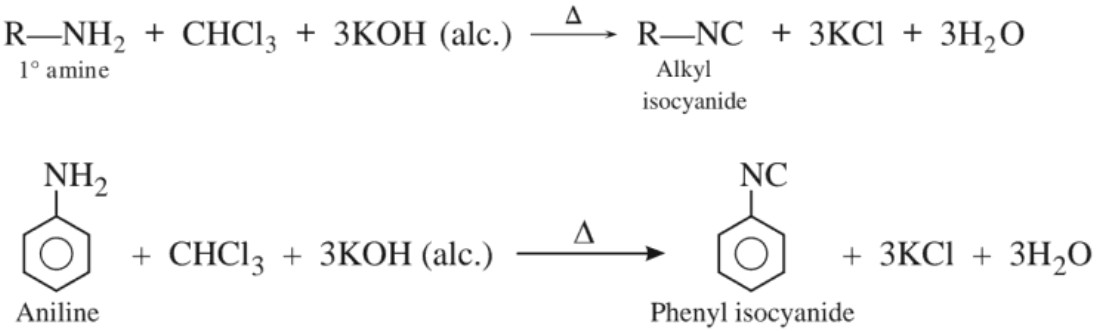
(c) Reaction with chloroform (Carbylamine reaction)
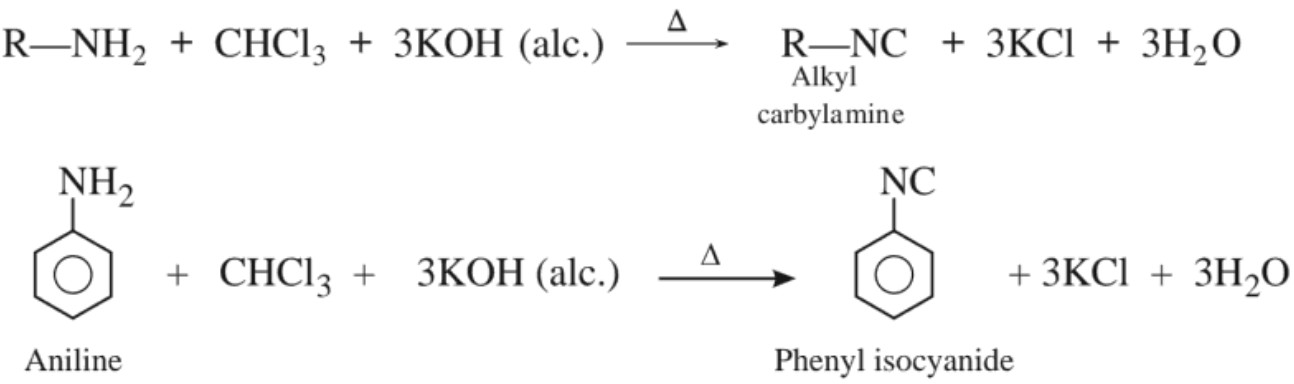
(d) Reaction with nitrous acid :
Reaction with nitrous acid helps in distinguishing between amines. Primary amines react with nitrous acid to form alcohols.
Secondary amines react with nitrous acid to form a yellow green oily layer of N – nitrosoamines. N – Nitrosoamines on warming with a crystal of phenol and a few drops of conc. H2SO4 form green solution which when treated with aqueous NaOH, turns deep blue and then red on dilution. This reaction is called Liebermann’s nitroso reaction.
tert – Amines readily dissolve in nitrous acid forming crystalline trialkyl ammonium nitrite.
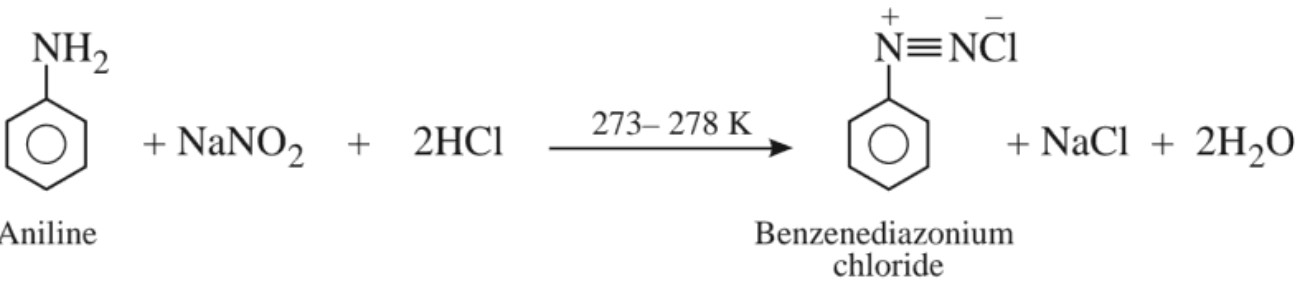
(e) Diazotization :
(f) Electrophlic substitution reactions :
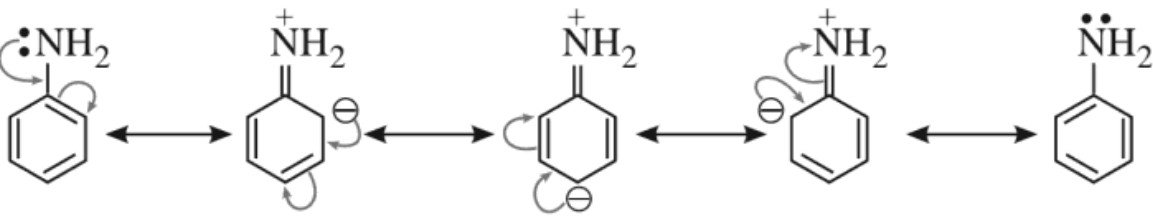
Due to resonance, electron density increases at ortho and para positions as compared to meta positions. Therefore, −NH2 group directs the incoming group to ortho and para positions.
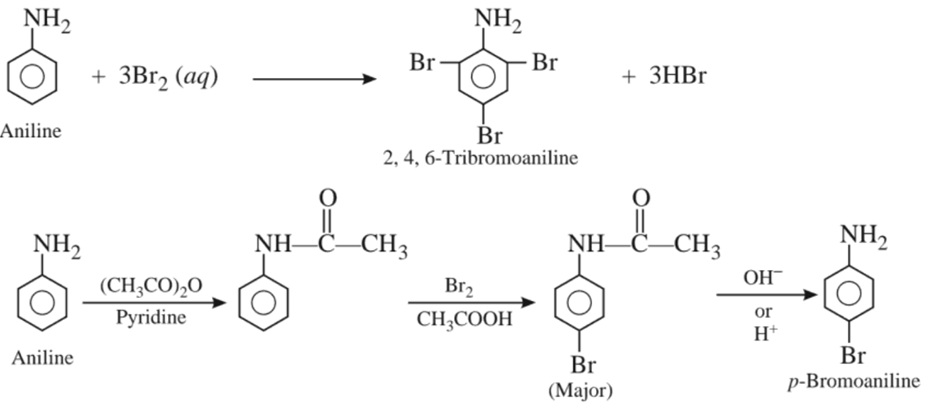
(i) Bromination :
(ii) Nitration :
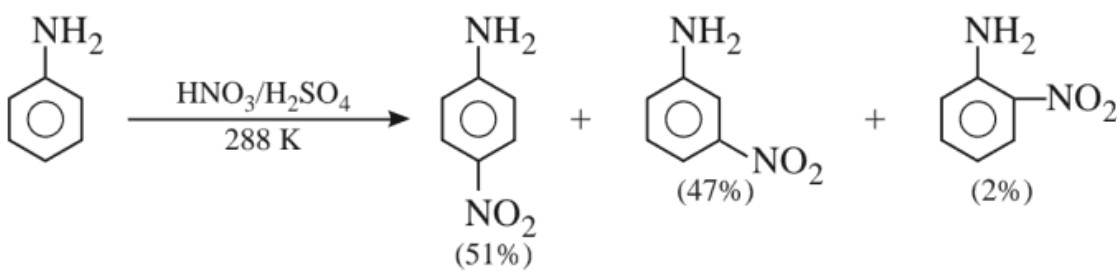
In strongly acidic medium, significant amount of meta isomer is obtained. This is due to the formation of anilinium ion which is meta directing. However, the p – nitro derivative can be obtained as the major product by protecting the –NH2 group by acetylation reaction.
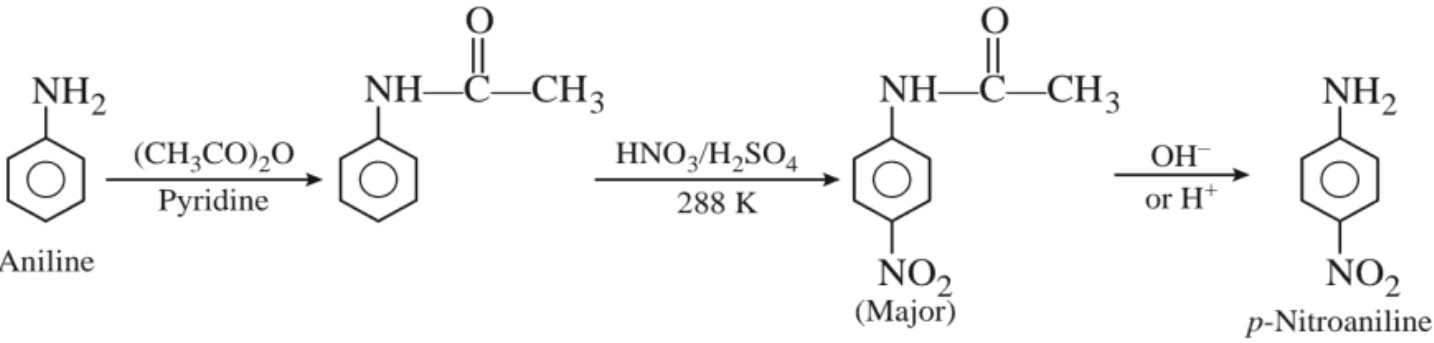
(iii) Sulphonation :
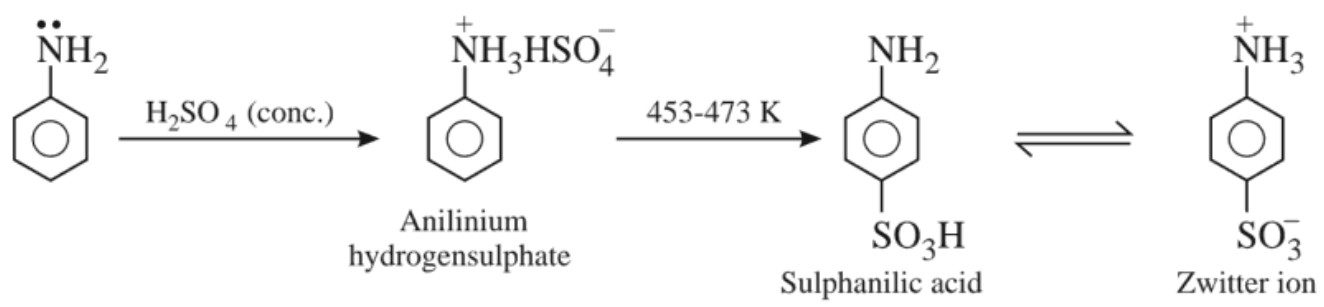
(iv) Friedel – Crafts reactions:

Due to salt formation, nitrogen of aniline acquires positive charge and thus acts as a strong deactivating group and hence does not allow Friedel – Crafts reaction to occur.
4
Preparation and properties of Aniline
Answer
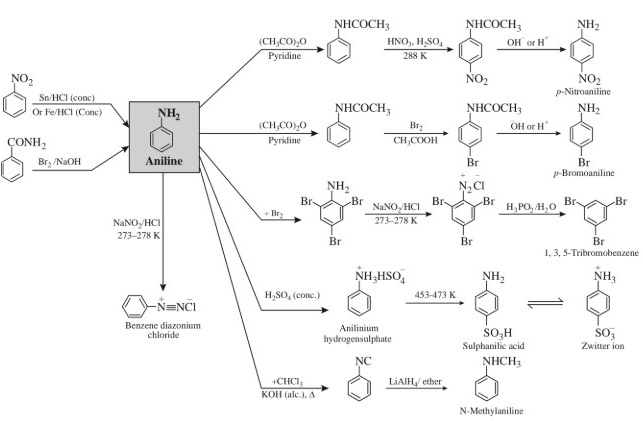
5
Diazonium Salts
Answer
(a) General formula : RN+2X−Where R stands for an aryl group and X– ion may be Cl– , Br– , HSO–4 , BF–4 , etc. The N2(−N ≡ N) is called the diazonium group.
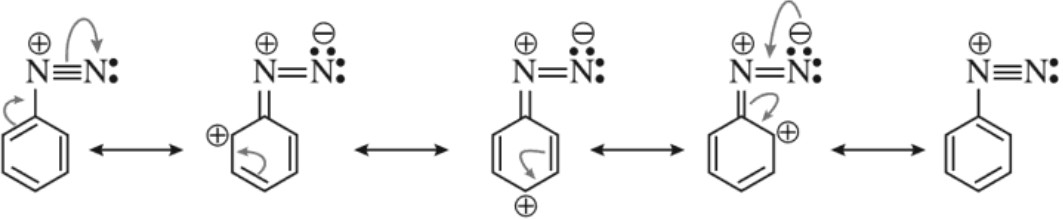
(b) Stability of diazonium salts :
Arenediazonium salts are much more stable than alkyl diazonim salts. The stability of arene diazonium salt is due to the dispersal of the positive charge over the benzene ring as shown below.
(c) Preparation of diazonium salts
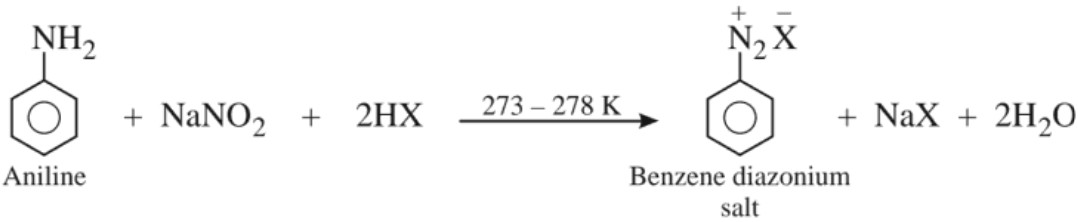
This process of conversion of a primary aromatic amine into its diazonium salt is called diazotization.
6
Chemical Properties of Diazonium Salts
Answer
(a) Important Synthetic Applications of Diazonium Chloride :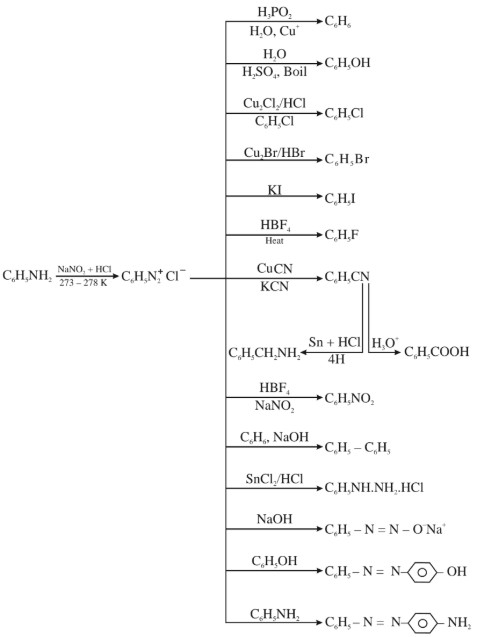
(b) Coupling reaction :
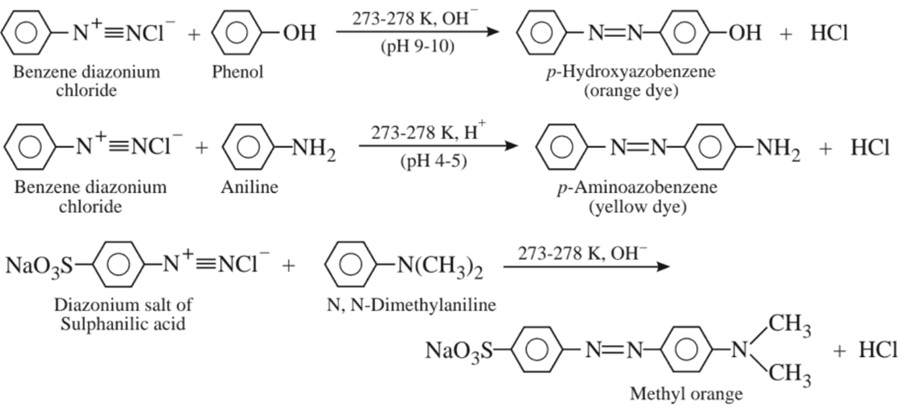
7
Some Important Name Reactions
Answer
(a) Gabriel phthalimide synthesis :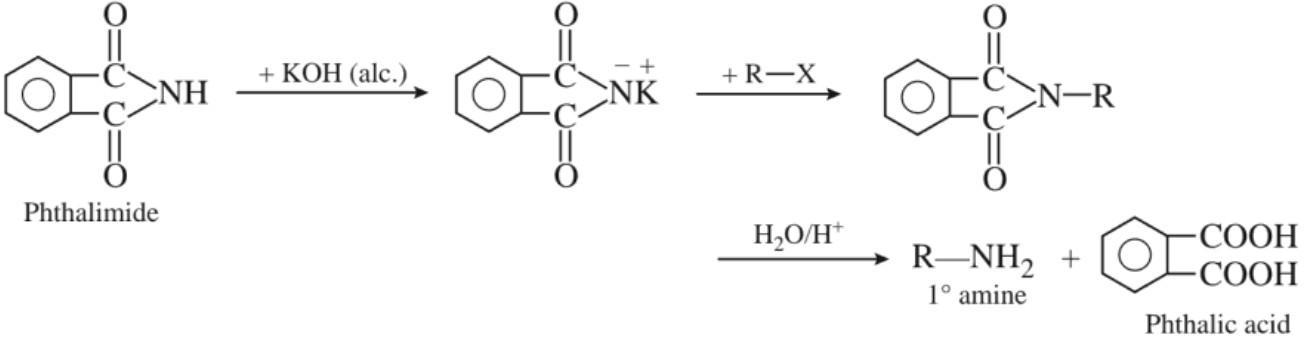
(b) Hoffmann bromamide reaction :
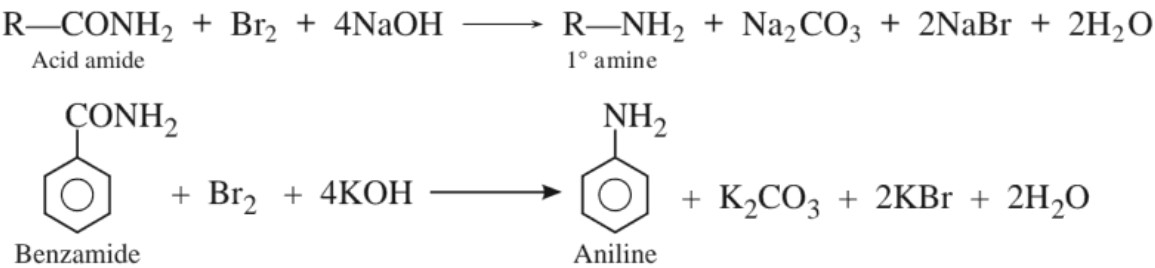
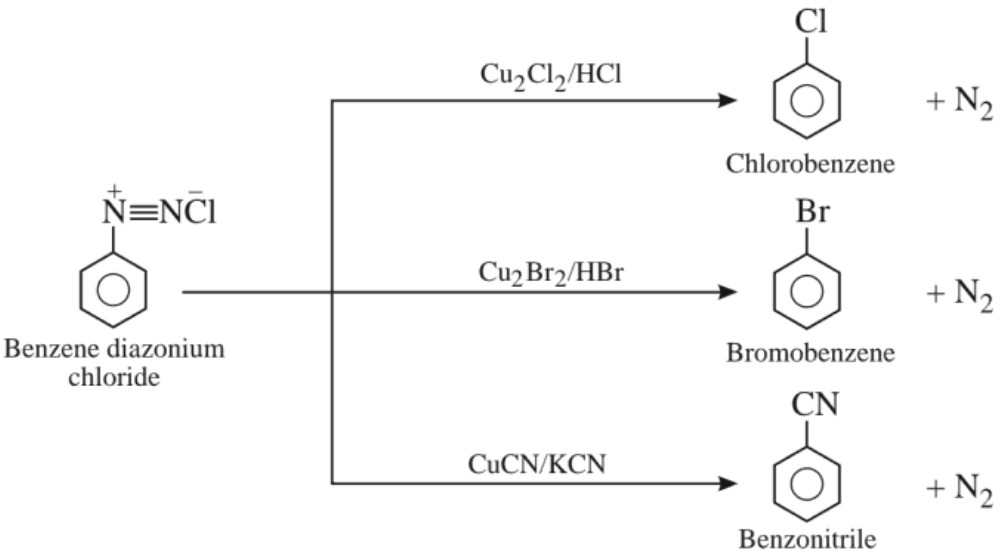
(c) Sandmeyer’s reaction :
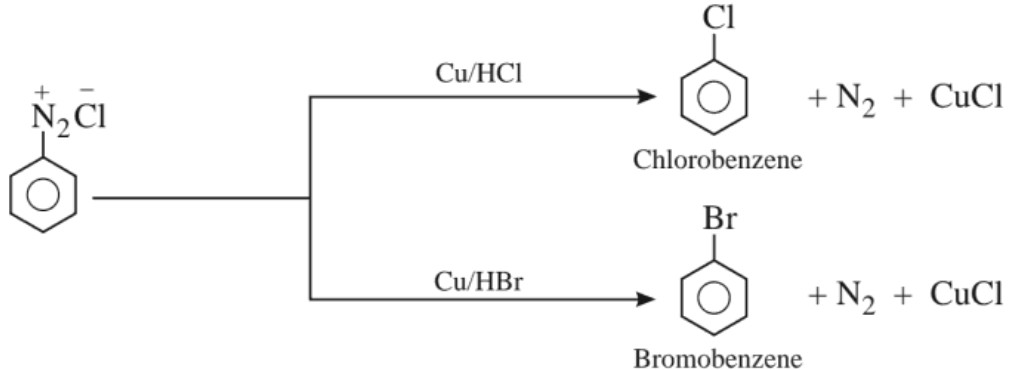
(d) Gatterman’s reaction :
(e) Carbylamine reaction (Isocyanide test) :
8
Test for Amines
Answer
