NCERT Revision Notes for Chapter 7 Alcohols, Phenols and Ethers Class 12 Chemistry
CBSE NCERT Revision Notes1
Answer
Alcohols : −OH group (−ol) : ROHPhenol :
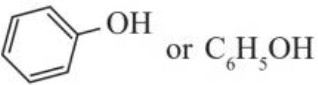
Classification of alcohol : 1° , 2°, 3° alcohols
2
Answer
Alcohol : R–O–H : sp3 hybridised orbital of C is overlapping with sp3 hybridised orbital of oxygen forming σ – bond.Phenol : C – O bond is sp2 hybridised i.e. partial double bond character due to resonance. It causes decrease in C – O bond length in phenol as compared to alcohols.
e– density is more at o and p – position for electrophilic attack.
3
Answer
I. From alkenes :
(a) From acid catalysed hydration (using Markovnikov rule)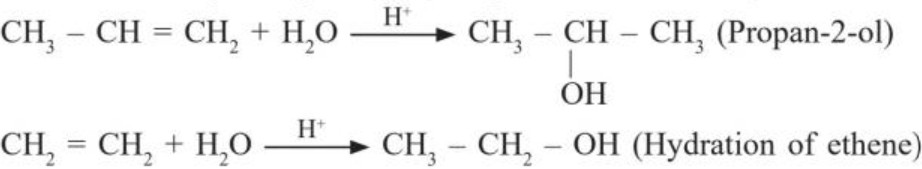
(b) Hydroboration – oxidation reaction : Diborane (B2H6) reacts with alkenes to give trialkyl boranes as addition product. This is oxidised to acohol by H2O2 in the presence of aqueous sodium hydroxide. In the reaction, Anti – Markovnikov product is obtained in high yield . 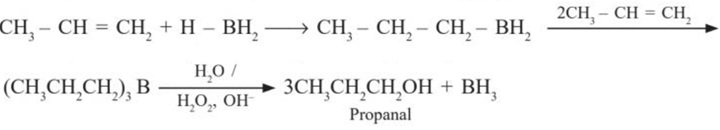
II. Reduction : 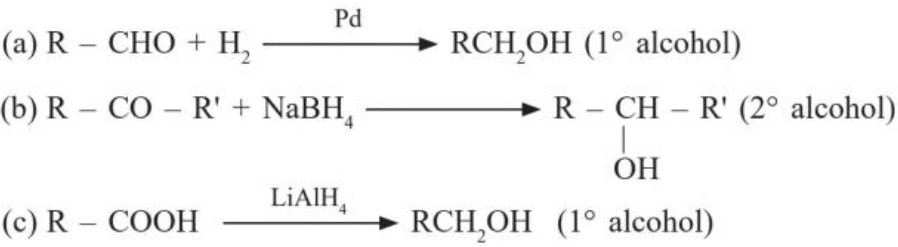
III. From Grignard reagent : 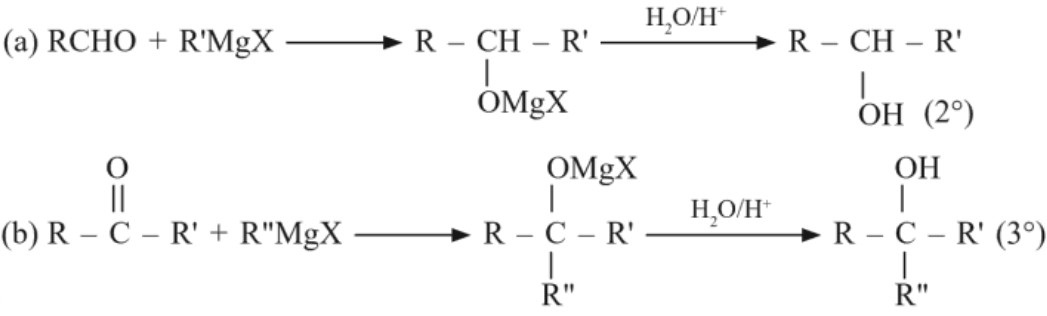
IV. Hydrolysis :
(a) R – X + aq KOH → R – OH + KX ![]()
4
Answer
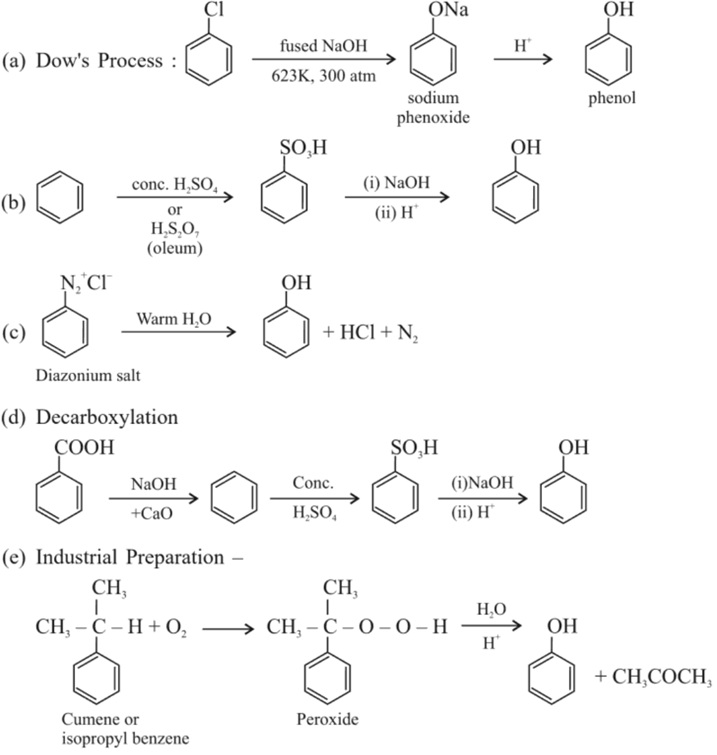
5
Answer
(a) Boiling point – increases with increase in number of carbon atoms due to increase in van der Waals’ forces of attraction. Phenols and alcohols have intermolecular hydrogen bonding due to which they have more boiling points than other compounds of comparable molar masses.(b) Solubility : They are soluble in water due to their ability to form intermolecular hydrogen bonds with water molecules.
6
Answer
Alcohols can act as nucleophile as well as electrophile.I. Reactions involving C – O bond cleavage :
Lucas reagent = Concentrated HCl + anhydrous ZnCl2
3° alcohol : turbidity appears immediately,
2° alcohol : turbidity appears after five minutes,
1° alcohol : turbidity appears on heating.
(c) Dehydration : Removal of water (I Method):
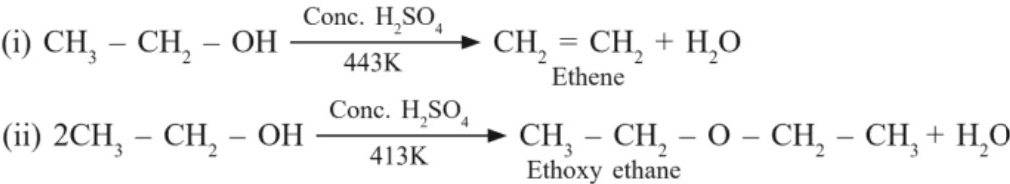
(d) Oxidation : (Dehydrogenation = loss of hydrogen)

(ii) Mild oxidation : CrO3 or PCC or Cu/573K [PCC = pyridinium chlorochromate]
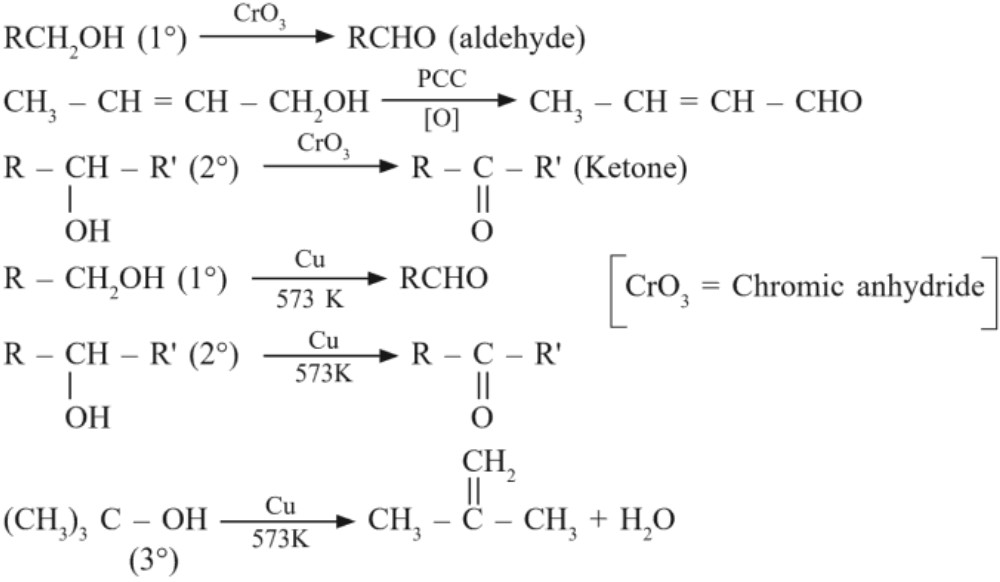
II. Reactions involving cleavage of O – H bond :
(a) Acidity of alcohols and phenols and reactions with metals:
2ROH + 2Na → 2RONa + H2
• Order of acidity of alcohols (acidic strength):
(i) Electron releasing groups increase electron density on oxygen tending to decrease the polarity of –OH bond. This decreases the acid strength.
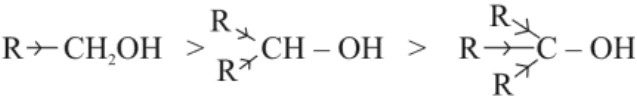
(ii) Phenol is more acidic than alcohol because
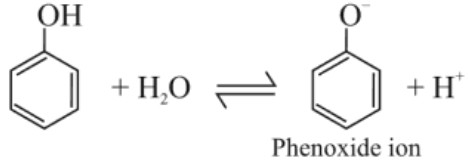
In alkoxide ion, negative charge is localised on oxygen while in phenoxide ion, charge is delocalised due to which phenoxide ion is more stable and favours ionisation of phenol.
(iii) Electron withdrawing group like –NO2 increase the acidic strength of phenol mainly at ortho and para – positions. It is due to the effective delocalisation of negative charge in phenoxide ion.
While electron releasing groups like alkyl groups do not favour the formation of phenoxide ion resulting in decrease in acidic strength.
(b) Esterification : Water should be removed as soon as possible so that reversible reaction would not occur.
7
Answer
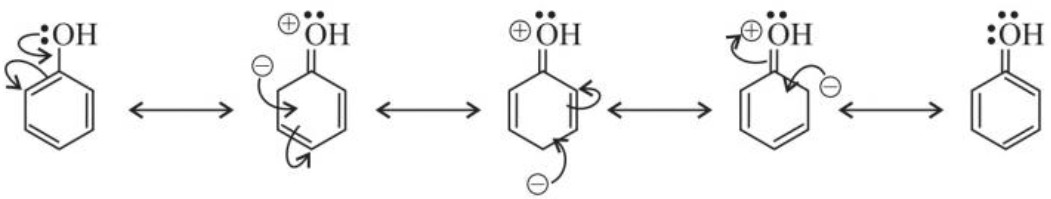
(a) Electrophilic substitution reaction : 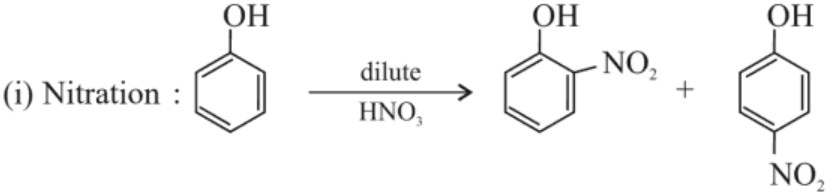
NOTE : Ortho – nitro phenol is more steam volatile than p – nitro phenol because p – nitro phenolis in association due to intermolecular hydrogen bonding which is stable than intramolecular hydrogen bonding of o – nitro phenol. 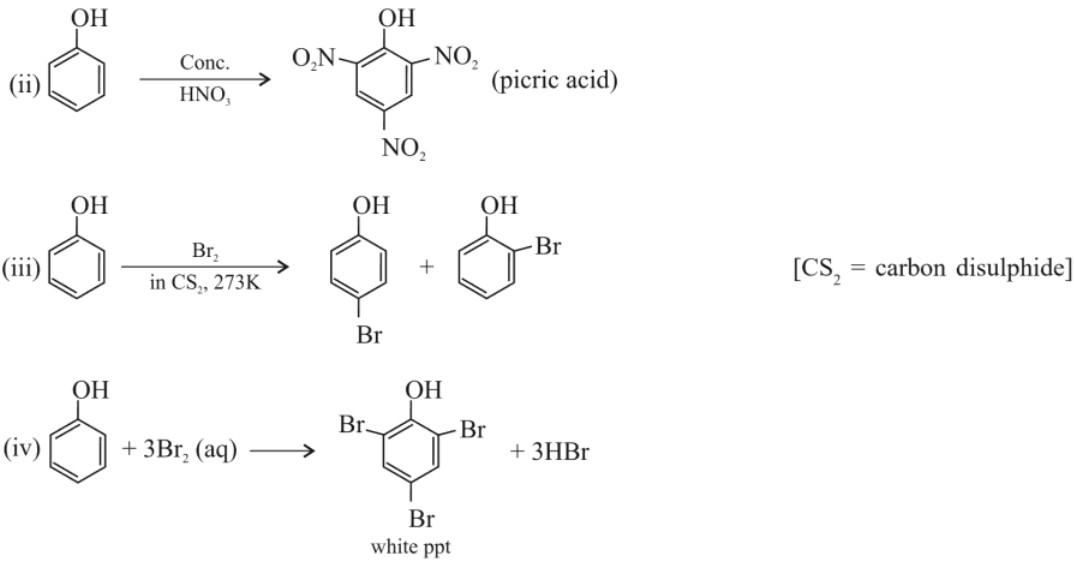
(b) Kolbe’s reaction : 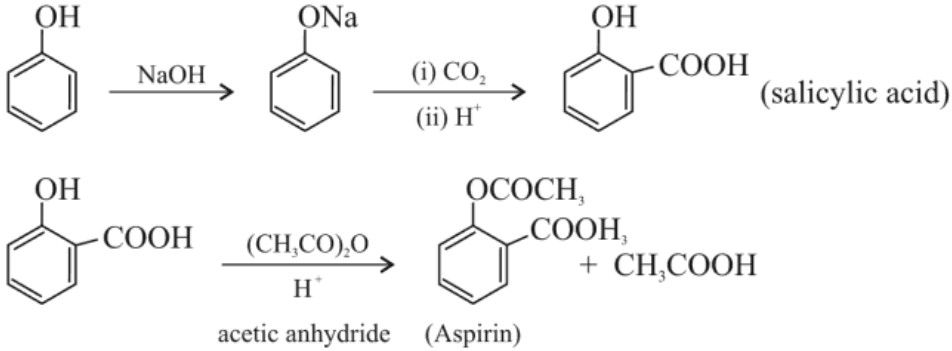
(c) Reimer – Tiemann reaction : 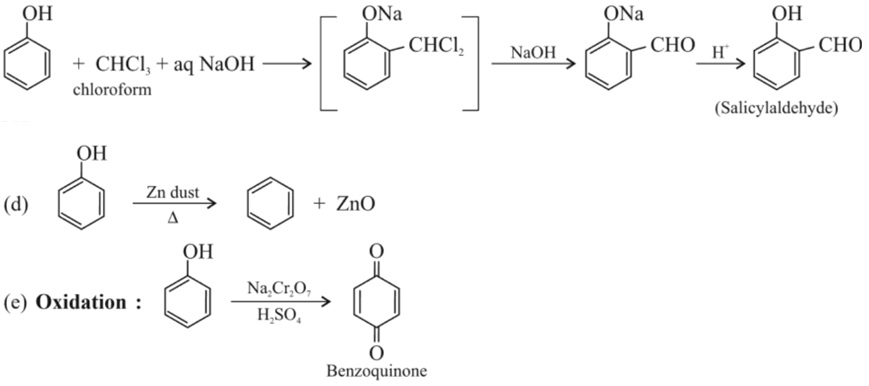
(f) Chemical test for phenols :
(i) Bromine water test 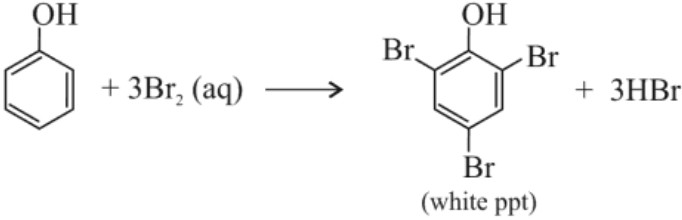
(ii) anhydrous FeCl3 test: 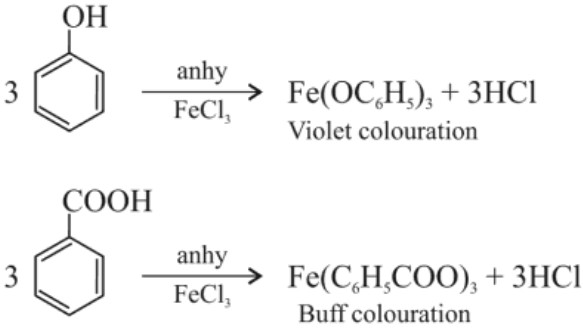
(iii) Azo dye test or coupling reaction : 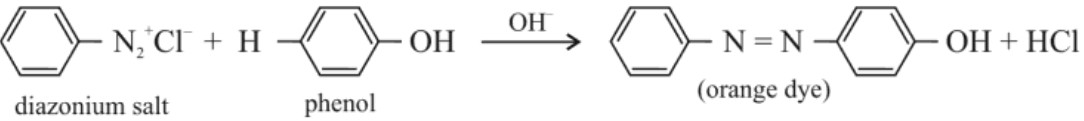
8
Answer
(a) Methanol (Wood spirit) : Highly poisonous – used in denaturation of alcohol and making paints.Preparation :
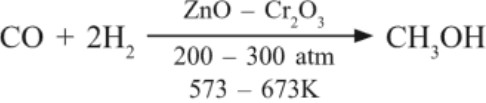
(b) Ethanol : Prepared by fermentation of sugar
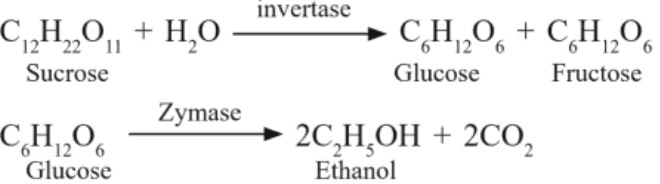
(c) Denatured alcohol : To prevent the misuse of alcohol in industries, few poisonous substances are added in alcohol, for e.g. methanol, CuSO4. Pyridine. Such an alcohol is known as denatured alcohol.
9
Answer
(a) Preparation of ethers:(b) Williamson’s synthesis : for the preparation of symmetrical and unsymmetrical ethers.
R’O– acts as a nucleophile (alkoxide ion) and attacks on primary alkyl halide by SN2 mechanism.
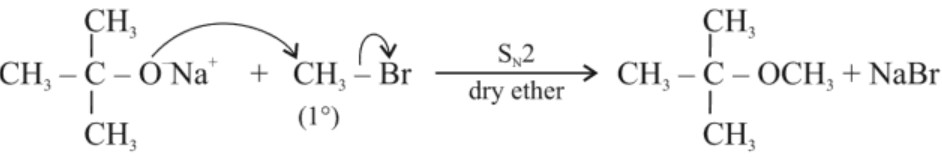
Limitation : Alkyl halide must be primary. If secondary and tertiary alkyl halides are used, elimination competes over substitution and alkene is formed.
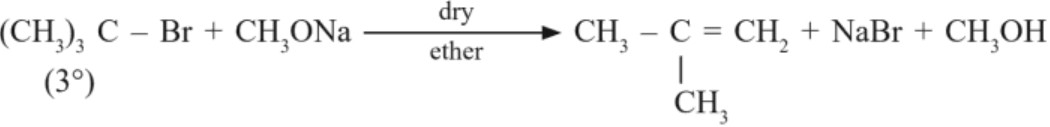
Because RO– (alkoxide) is not only a nucleophile but a strong base as well. It reacts with alkyl halides leading to elimination reaction.
10
Answer
(a) ROR’ + HX → RX + R’OH,(i) CH3CH2OCH3 + HI → CH3CH2OH + CH3I [ Conc. HI or HBr at high T]
CH3CH2OCH3 + HI(excess) → CH3CH2I + CH3I + H2O
(ii) When one of the alkyl group is 3°, halide formed is tertiary halide.
(CH3)3C – O – CH3 + HI → (CH3)3 C – I + CH3OH
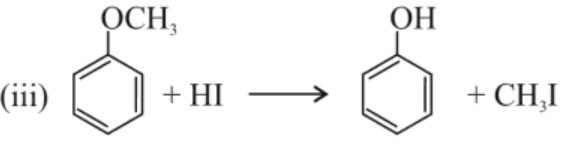
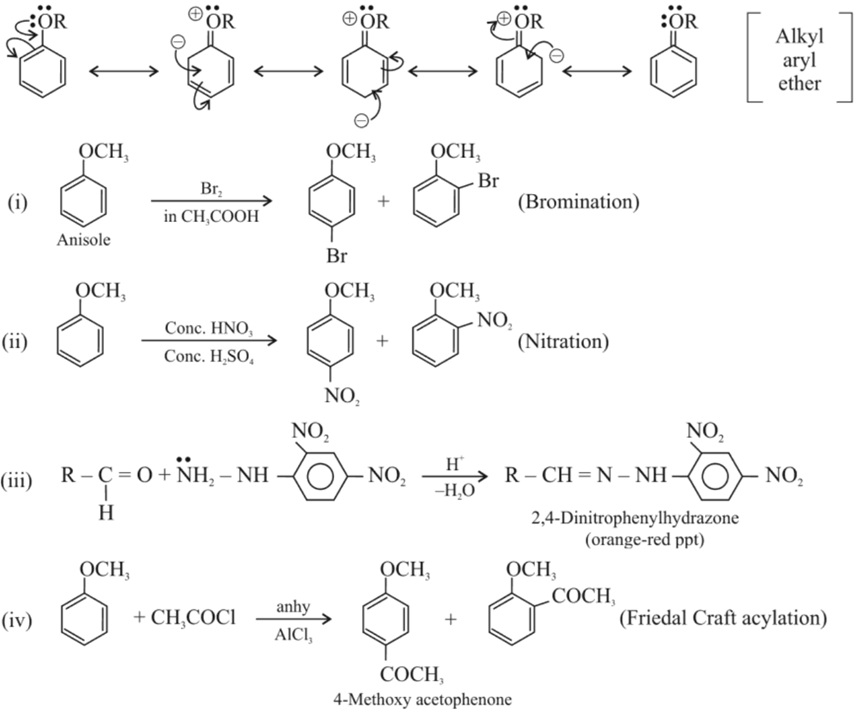
(b) Electrophilic substitution reaction : −OR group is o, p–directing and activates the benzene ring towards electrophilic substitution reacton.