VSAQ for Chapter 2 Exploring Substances: Acidic, Basic and Neutral Class 7 Science NCERT
Important Questions1
Q1: The substances used to test whether a substance is acidic or basic are known as ________
Answer
The substances used to test whether a substance is acidic or basic are known as Indicators
VSAQ
2
Q2: Name the most commonly used indicator.
Answer
Litmus paper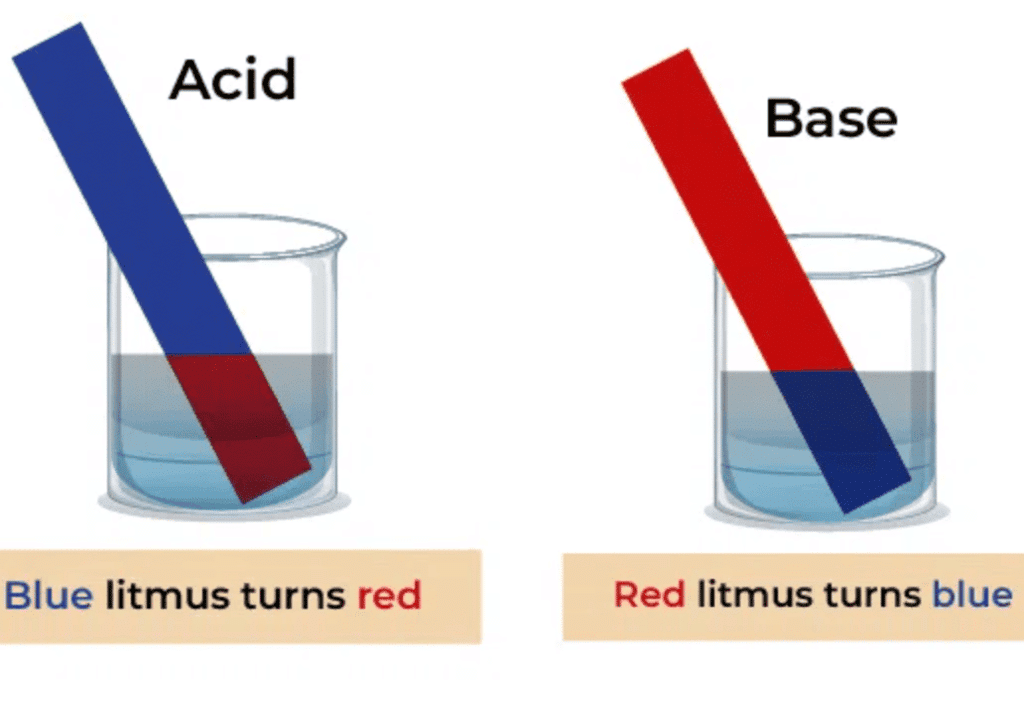
VSAQ
3
Q3: From where do we extract litmus to be used as an indicator?
Answer
Lichens
VSAQ
4
Q4: In acidic solution, litmus paper turns into ______
Answer
In acidic solution, litmus paper turns into RedLitmus paper is a pH indicator that turns red in acidic solutions, indicating the presence of acids.
VSAQ
5
Q5: In basic solution, litmus paper turns into ____
Answer
In basic solution, litmus paper turns into BlueLitmus paper changes to blue in basic solutions, indicating the presence of bases.
VSAQ
6
Q6: In distilled water, litmus paper turns into _________
Answer
In distilled water, litmus paper turns into purpleLitmus paper remains purple in neutral distilled water, as it is neither acidic nor basic.
VSAQ
7
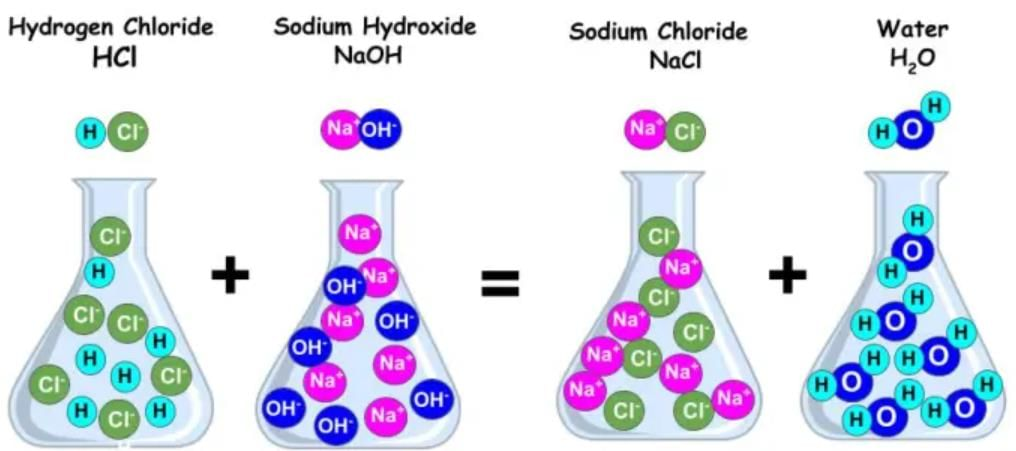
Q7: The reaction between an acid and a base is known as _________
Answer
The reaction between an acid and a base is known as neutralization.Neutralization is the chemical reaction that occurs when an acid and a base react to form water and a salt.
VSAQ
8
Q8: Red rose indicator turns acidic solutions _______
a. Dark pink
b. Purple
c. Blue
d. None of these
a. Dark pink
b. Purple
c. Blue
d. None of these
Answer
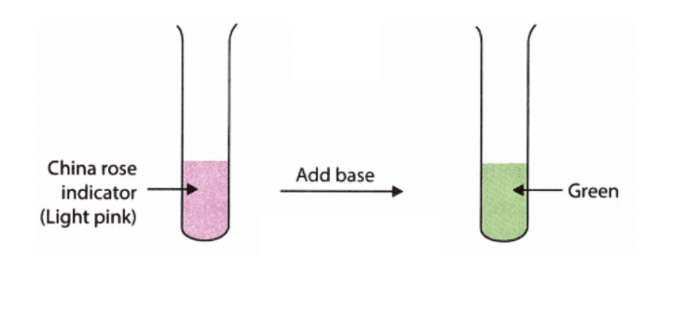
a. Dark pinkChina rose indicator changes to dark pink in acidic solutions, indicating their acidic nature.
VSAQ
9
Q9: Red/ China rose indicator turns basic solutions to ______
a. Dark pink
b. Purple
c. Blue
d. Green
a. Dark pink
b. Purple
c. Blue
d. Green
Answer
d. GreenThe China rose indicator changes the colour of basic solutions to green.
In contrast, it turns acidic solutions to dark pink.
VSAQ
10
Q10: Salt and water are produced in the neutralization process with the evolution of ____________
Answer
Salt and water are produced in the neutralization process with the evolution of Heat
VSAQ
11
Q11: Name the acid present in our stomach.
Answer
HCL is present in our stomach.Hydrochloric acid helps break down food and activates digestive enzymes, facilitating the digestion process.
VSAQ
12
Q12: Name the acid present in the sting of an ant.
Answer
Formic acid is present in the sting of an ant.
VSAQ
13
Q13: State the nature of the soap solution.
Answer
Basic
VSAQ
14
Q14: State the nature of baking soda.
Answer
Basic
VSAQ
15
Q15: State the nature of lemon juice.
Answer
Acidic
VSAQ
16
Q16: Why does lemon juice and orange juice taste sour?
Answer
Because they contain acids.
VSAQ
17
Q17: Why does baking soda taste bitter?
Answer
Because it is basic in nature.
VSAQ
18
Q18: State one property of acids.
Answer
Acids are sour in taste.
VSAQ
19
Q19: State one property of bases.
Answer
Bases are bitter in taste.
VSAQ
20
Q20: Tina rubs a solution between her fingers and feels soapy; what is the nature of that solution?
Answer
Basic
VSAQ
21
Q21: Complete the following reaction:
HCl + NaOH ----> ______ + H2O
HCl + NaOH ----> ______ + H2O
Answer
NaCl
VSAQ
22
Q22: Ammonia is found in many household products, such as window cleaners. It turns red litmus blue. Its nature _______________
Answer
Ammonia is found in many household products, such as window cleaners. It turns red litmus blue. Its nature is Basic
VSAQ
23
Q23: The wastes of many factories contain____________
Answer
The wastes of many factories contain Acids
VSAQ
24
Q24: Blue litmus paper is dipped in a solution. It remains blue, what is the nature of the solution?
Answer
Basic
VSAQ
25
Q25: Hydrogen ion is common to all acids. True/False
Answer
True
VSAQ
26
Q26: Name a base that is also used in soda acid fire extinguishers.
Answer
Baking soda.
VSAQ
27
Q27: Aqueous solution of acid conducts electricity due to ______________ present in it.
Answer
ion
VSAQ
28
Q28: Define pH.
Answer
pH is the measure of the Acidity or Alkalinity of a solution. The term pH stands for a negative log of hydrogen ion concentration.
VSAQ
29
Q29: The bases that dissolve in water are known as alkalies. True/ False.
Answer
True
VSAQ
30
Q30: Phenolphthalein becomes colourless in __________ and pink in_________.
Answer
Colourless in acid and pink in base.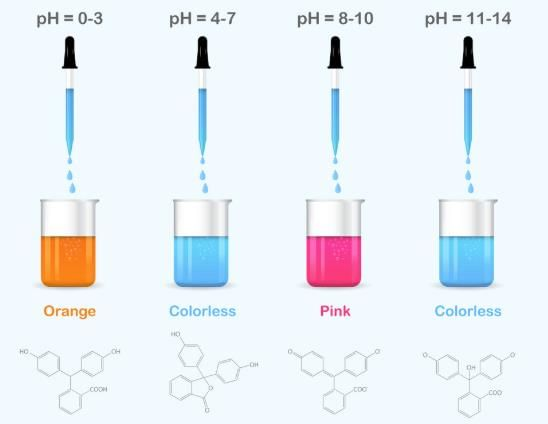
Phenolphthalein
VSAQ
31
Q31: Name the acid present in vinegar.
Answer
Acetic acid
VSAQ
32
Q32: Name the acid present in lemon.
Answer
citric acid
VSAQ
33
Q33: Change of colour in an acid or base depends on
a. Type of indicator
b. The acidic property of that substance
c. Basic property of that substance
d. None of these
a. Type of indicator
b. The acidic property of that substance
c. Basic property of that substance
d. None of these
Answer
Type of indicator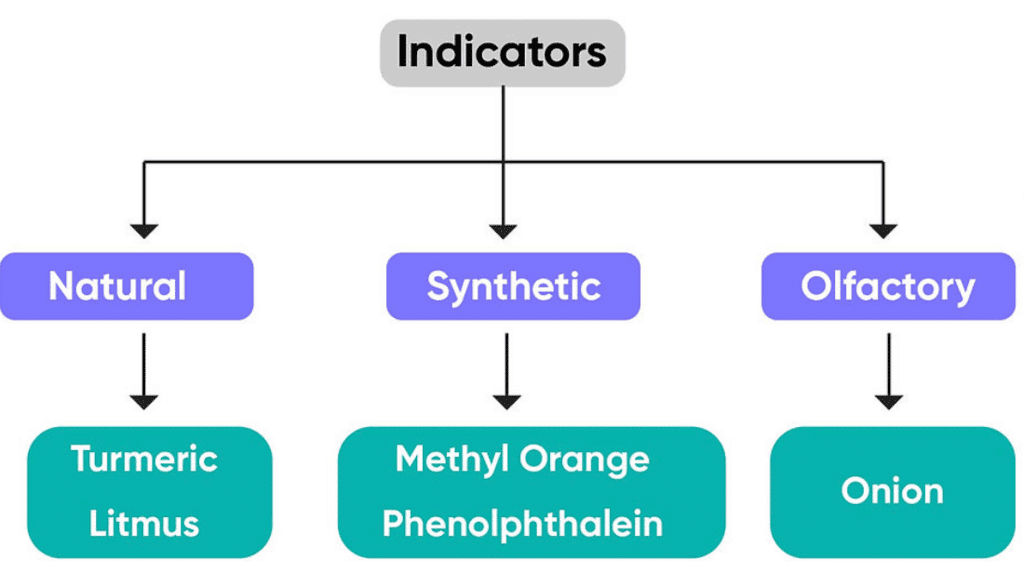
VSAQ
34
Q34: Name the acid present in curd.
Answer
Lactic acid
VSAQ